A) 2-Chloro-4-isopropyl-2,6-dimethyloctane
B) 2-Chloro-4-isopropyl-2,7-dimethylnonane
C) 2,6-Dimethyl-2-chloro-4-isopropyloctane
D) 7-Chloro-5-isopropyl-3,7-dimethyloctane
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following halides in order of decreasing polarity,putting the most polar first. 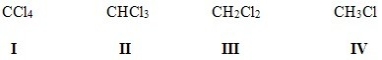
A) II > IV > I > III
B) IV > II > I > III
C) I > II > III > IV
D) IV > III > II > I
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 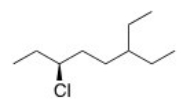
A) (R) -3-Chloro-6-ethyloctane
B) (S) -3-Chloro-6-ethyloctane
C) (S) -6-Chloro-3-ethyloctane
D) (R) -6-Chloro-3-ethyloctane
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the reactions of alkyl halides is true?
A) The characteristic reactions of alkyl halides are addition and elimination.
B) The characteristic reactions of alkyl halides are addition and substitution.
C) The characteristic reactions of alkyl halides are elimination and substitution.
D) The characteristic reactions of alkyl halides are oxidation and reduction.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the SN2 mechanism for nucleophilic substitution reactions is true?
A) Involves one step and occurs with retention of configuration.
B) Involves two steps and occurs with inversion of configuration.
C) Involves one step and occurs with inversion of configuration.
D) Involves one step and occurs with racemization.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements explain why aryl halides and vinyl halides do not undergo nucleophilic substitution by either the SN1 or SN2 mechanism?
A) They don't undergo SN1 reactions because a higher percent s-character makes the bond longer and stronger.
B) They don't undergo SN2 reactions because a higher percent s-character makes the bond shorter and stronger.
C) They don't undergo SN2 reactions because heterolysis of the C-X bond forms a highly unstable carbocation.
D) They don't undergo SN1 reactions because the carbocation is highly electronegative.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides is a secondary alkyl halide? 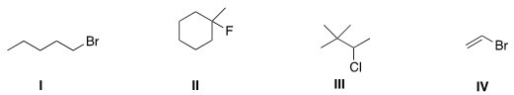
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a characteristic of an SN1 reaction?
A) The rate is proportional to the concentration of substrate.
B) The reaction is favored in polar protic solvents.
C) The rate is independent of the concentration of the nucleophile.
D) The electrophilic carbon undergoes inversion of stereochemistry.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true?
A) In polar protic solvents,nucleophilicity decreases down a column of the periodic table as the size of the anion increases.
B) Nucleophilicity is affected by the solvent used in a substitution reaction.
C) Polar protic solvents are capable of intermolecular hydrogen bonding.
D) Polar protic solvents solvate both cations and anions.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solvents is not a polar protic solvent? 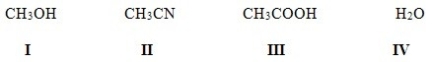
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following carbocations in order of decreasing stability,putting the most stable first. 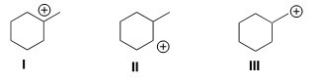
A) I > II > III
B) II > I > III
C) III > I > II
D) III > II > I
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 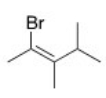
A) (E) -2-Bromo-3,4-dimethyl-2-pentene
B) (Z) -1-Bromo-1,2,3-trimethyl-1-butene
C) (Z) -2-Bromo-3,4-dimethyl-2-pentene
D) (E) -1-Bromo-1,2,3-trimethyl-1-butene
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 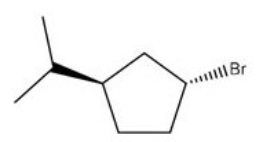
A) (1R,3R) -1-bromo-3-isopropylcyclopentane
B) (1R,3S) -1-bromo-3-isopropylcyclopentane
C) (1S,3R) -1-bromo-3-isopropylcyclopentane
D) (1S,3S) -1-bromo-3-isopropylcyclopentane
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pick the solvent that gives the fastest SN2 reaction between CH2CH2Br and -OCH3.
A) CH2CH2OH
B) CH3OH
C) DMSO
D) H2O
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the nucleophilic substitution reaction shown below? 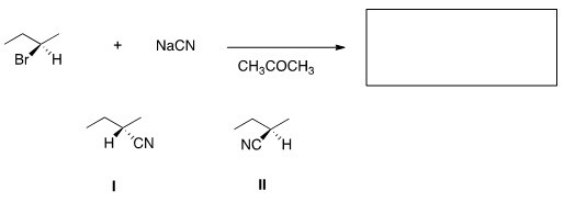
A) Only I
B) Only II
C) I and II
D) None
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of bromoethane with sodium acetate affords the substitution product methyl acetate.What is the effect of doubling the concentration of sodium acetate on the rate of the reaction?
A) The rate remains the same.
B) The rate decreases by a factor of 2.
C) The rate increases by a factor of 2.
D) The rate increases by a factor of 4.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides is a primary alkyl halide? 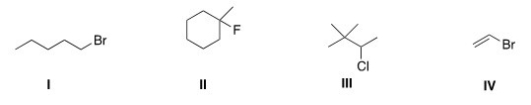
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true?
A) Sodium ethoxide is a better nucleophile than sodium tert-butoxide.
B) Sodium tert-butoxide and sodium ethoxide have similar strengths as bases.
C) Sterically hindered bases are also called nonnucleophilic bases.
D) Steric hindrance decreases basicity but not nucleophilicity.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of increasing nucleophilicity,putting the least nucleophilic first. 
A) II < IV < I < III
B) IV < III < II < I
C) III < IV < I < II
D) II < I < IV < III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction,use the identity of the alkyl halide and nucleophile to determine which substitution mechanism occurs.Then determine which solvent affords the faster reaction. 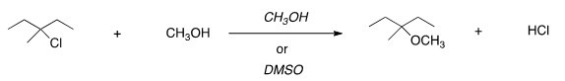
A) SN1,CH3OH
B) SN1,DMSO
C) SN2,CH3OH
D) SN2,DMSO
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 73
Related Exams