A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Rank the following in order of increasing leaving group ability,putting the worst leaving group first. 
A) IV < II < III < I
B) III < IV < I < II
C) II < IV < I < III
D) I < III < II < IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 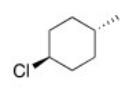
A) trans-4-Methylcyclohexyl chloride
B) trans-p-Chloromethylcyclohexane
C) trans-4-Methyl-1-chlorocyclohexane
D) trans-1-Chloro-4-methylcyclohexane
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures have the correct common name? 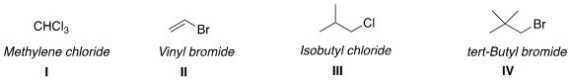
A) I and II
B) II and III
C) I and III
D) III and IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the nucleophile and type of substitution reaction in the following: 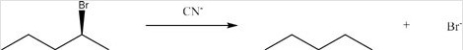
A) CN-,Sn1
B) CN-,Sn2
C) Br-,Sn1
D) Br-,Sn2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of increasing nucleophilicity,putting the least nucleophilic first. 
A) II < IV < I < III
B) IV < III < II < I
C) III < IV < I < II
D) II < I < IV < III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true?
A) All good leaving groups are weak bases with strong conjugate acids.
B) Left-to-right across a row of the periodic table,leaving group ability increases.
C) Down a column of the periodic table,leaving group ability increases.
D) Equilibrium favors the products of the nucleophilic substitution when the leaving group is a stronger base than the nucleophile.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 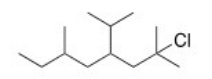
A) 2-Chloro-4-isopropyl-2,6-dimethyloctane
B) 2-Chloro-4-isopropyl-2,7-dimethylnonane
C) 2,6-Dimethyl-2-chloro-4-isopropyloctane
D) 7-Chloro-5-isopropyl-3,7-dimethyloctane
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Rank the following molecules in order of increasing polarity,putting the least polar first. 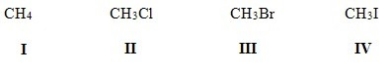
A) IV < III < II < I
B) I < IV < III < II
C) I < II < III < IV
D) II < I < III < IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of decreasing leaving group ability,putting the best first. 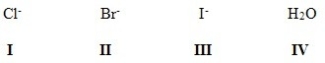
A) III > II > IV > I
B) III > II > I > IV
C) IV > I > II > III
D) II > III > I > IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 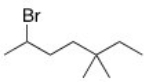
A) 2-Bromo-5,5-dimethylheptane
B) 3,3-Dimethyl-6-bromoheptane
C) 6-Bromo-3,3-dimethylheptane
D) 2-Bromo-5,5-dimethyloctane
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction of what nucleophile and substrate is represented by the following transition state? 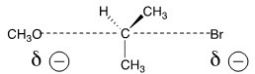
A) Methanol with 2-bromopropane
B) Methoxide with 2-bromopropane
C) Methoxide with 1-bromopropane
D) Methanol with 1-bromopropane
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following alkyl halides would react the fastest with H2O in SN1 reaction? CH3CH2CH2CH2Br,(CH3) 2CHCH2Br,CH3CH2CH(CH3) Br,(CH3) 3CBr
A) CH3CH2CH2CH2Br
B) (CH3) 2CHCH2Br
C) CH3CH2CH(CH3) Br
D) (CH3) 3CBr
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the rate-determining step of an SN1 reaction mechanism?
A) Reaction of the nucleophile with the carbocation to form the product.
B) Breaking the bond between the carbon and the leaving group to generate a carbocation.
C) Attack of the nucleophile on the substrate to generate a pentavalent carbon.
D) None of these.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pick the solvent that gives the fastest SN2 reaction between CH2CH2Br and -OCH3.
A) CH2CH2OH
B) CH3OH
C) DMSO
D) H2O
F) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following is a polar aprotic solvent? 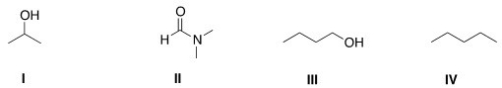
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction,use the identity of the alkyl halide and nucleophile to determine which substitution mechanism occurs.Then determine which solvent affords the faster reaction. 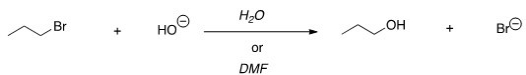
A) SN1,H2O
B) SN1,DMF
C) SN2,H2O
D) SN2,DMF
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 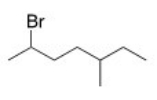
A) 2-Bromo-5-methyloctane
B) 2-Bromo-3-methylheptane
C) 2-Bromo-5-methylheptane
D) 6-Bromo-3-methylheptane
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 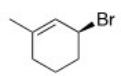
A) (R) -3-Bromo-1-methylcyclohexene
B) (S) -3-Bromo-1-methylcyclohexene
C) (S) -1-Bromo-3-methyl-2-cyclohexene
D) (R) -1-Bromo-3-methyl-2-cyclohexene
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC name of the following compound? 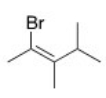
A) (E) -2-Bromo-3,4-dimethyl-2-pentene
B) (Z) -1-Bromo-1,2,3-trimethyl-1-butene
C) (Z) -2-Bromo-3,4-dimethyl-2-pentene
D) (E) -1-Bromo-1,2,3-trimethyl-1-butene
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 73
Related Exams