A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which C=O functional group is present in the following molecule? 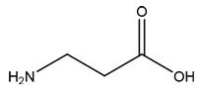
A) Carboxylic Acid
B) Ester
C) Ketone
D) Aldehyde
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do heteroatoms confer reactivity on a particular molecule?
A) Because they have lone pairs and create electron-rich sites on carbon.
B) Because they have lone pairs and create electron-deficient sites on carbon.
C) Because they are electronegative and act as electrophiles.
D) Because they are electropositive and act as nucleophiles.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the molecule atenolol (a β blocker used to treat hypertension) .Which of the following lists the correct functional groups present in atenolol? 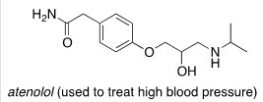
A) Primary alcohol,amide,primary amine,aromatic,ether
B) Secondary alcohol,amide,secondary amine,aromatic,ether
C) Secondary alcohol,amide,primary amine,aromatic,ether
D) Secondary alcohol,amide,secondary amine,aromatic,ester
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a tertiary amine? 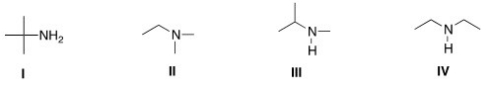
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do π bonds confer reactivity on a particular molecule?
A) Because π bonds are difficult to break in chemical reactions.
B) Because π bonds make a molecule an acid.
C) Because π bonds are easily broken in chemical reactions.
D) Because π bonds make a molecule an electrophile.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the intermolecular forces present in the following molecule: 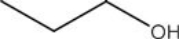
A) Van der Waals
B) Dipole-dipole interactions
C) Hydrogen bonding
D) More than one of these answer choices is correct.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated bond is: 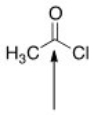
A) Nucleophilic because it is electron-deficient.
B) Electrophilic because it is electron-rich.
C) Nucleophilic because it is electron-rich.
D) Electrophilic because it is electron-deficient.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would be expected to be more soluble in hexane (C6H14) ? 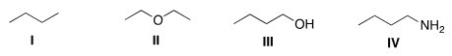
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following could most likely serve as an ionophore? 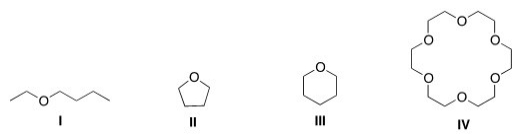
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the molecule donepezil (used to treat Alzheimer's disease) .Which of the following lists the correct functional groups present in donepezil? 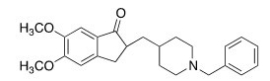
A) Amide,aromatic,ether,ketone
B) Amide,aromatic,ester,ketone
C) Amine,aromatic,ester,ketone
D) Amine,aromatic,ether,ketone
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing boiling point,putting the compound with the highest boiling point first. 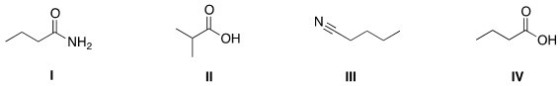
A) I > III > IV > II
B) IV > II > I > III
C) IV > I > II > III
D) I > IV > II > III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds can form intermolecular hydrogen bonds with a molecule similar to itself? 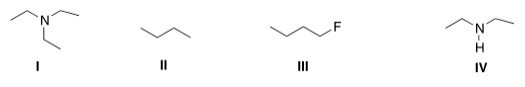
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures contains a primary amine? 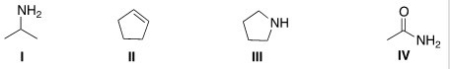
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 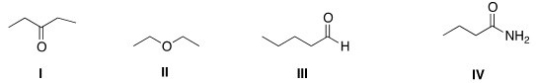
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which C=O functional group is present in the following molecule? 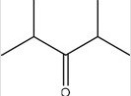
A) Carboxylic acid
B) Ester
C) Ketone
D) Aldehyde
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which C=O functional group is present in the following molecule? 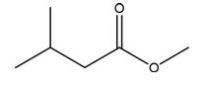
A) Carboxylic acid
B) Ester
C) Ketone
D) Aldehyde
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about vitamin A,drawn below,is true? 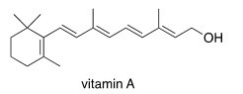
A) Vitamin A is soluble in H2O.
B) Vitamin A is insoluble in organic solvents.
C) Vitamin A contains an aromatic ring.
D) Vitamin A is insoluble in H2O.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the lowest boiling point? 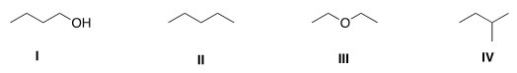
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 56
Related Exams