A) Mn in MnO2 is oxidized.
B) O in KClO3 is the oxidizing agent.
C) K in KClO3 is the reducing agent.
D) H in KOH is oxidized.
E) Cl in KClO3 is the reducing agent.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molarity of a 23.55-mL solution which contains 28.24 mg of sodium sulfate (used in dyeing and printing textiles, 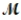 = 139.04 g/mol) .
= 139.04 g/mol) .
A) 8.625 M
B) 1.199 M
C) 0.8339 M
D) 0.2031 M
E) 0.008625 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances is the best electrolyte?
A) CO
B) CH3Cl
C) CH4
D) C2H5OH
E) HCl
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During a titration, the following data were collected. A 25 mL portion of an unknown acid solution was titrated with 1.0 M NaOH. It required 65 mL of the base to neutralize the sample. How many moles of acid are present in 3.0 liters of this unknown solution?
A) 2.6 moles
B) 0.9 moles
C) 8.7 moles
D) 7.8 moles
E) 5.6 moles
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances, when dissolved in water at equal molar concentrations, will give the solution with the lowest electrical conductivity?
A) CaCl2
B) HNO3
C) NH3
D) C6H12O6 (glucose)
E) CO2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g)
B) H2(g) + Cl2(g) → 2HCl(g)
C) 2H2O2(aq) → 2H2O(l) + O2(g)
D) Fe2O3(s) + 3H2SO4(aq) → Fe2(SO4) 3(aq) + 3H2O(l)
E) 2KMnO4(aq) + 10FeSO4(aq) + 8H2SO4(aq) → K2SO4(aq) + 2MnSO4(aq) + 5Fe2(SO4) 3(aq) + 8H2O(l)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrochloric acid is widely used as a laboratory reagent in refining ore for the production of tin and tantalum, and as a catalyst in organic reactions. Calculate the number of moles of HCl in 62.85 mL of 0.453 M hydrochloric acid.
A) 28.5 mol
B) 1.04 mol
C) 0.139 mol
D) 0.0285 mol
E) 0.00721 mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 1.25 L of 0.75 M nitric acid?
A) 0.60 mol
B) 0.75 mol
C) 0.94 mol
D) 1.7 mol
E) 1.9 mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2NaCl(l)  2Na(l) + Cl2(g)
2Na(l) + Cl2(g)
A) Acid-base
B) Precipitation
C) Combination
D) Displacement
E) Decomposition
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct set of products for the following reaction. Ba(OH) 2(aq) + HNO3(aq) →
A) BaN2(s) + H2O(l)
B) Ba(NO3) 2(aq) + H2O(l)
C) Ba(s) + H2(g) + NO2(g)
D) Ba2O(s) + NO2(g) + H2O(l)
E) No reaction occurs.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium carbonate, K2CO3, sodium iodide, NaI, potassium bromide, KBr, methanol, CH3OH, and ammonium chloride, NH4Cl, are soluble in water. Which produces the largest number of dissolved particles per mole of dissolved solute?
A) K2CO3
B) NaI
C) KBr
D) CH3OH
E) NH4Cl
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sodium ions are present in 325 mL of 0.850 M Na2SO4?
A) 1.66 × 1023 sodium ions
B) 3.33 × 1023 sodium ions
C) 4.99 × 1023 sodium ions
D) 6.20 × 1023 sodium ions
E) 1.57 × 1024 sodium ions
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of ions are released when 1.6 mol of ammonium phosphate, (NH4) 3PO4, is dissolved in water?
A) 0.40 mol
B) 1.6 mol
C) 3.2 mol
D) 4.8 mol
E) 6.4 mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium chloride, KCl, sodium sulfate, Na2SO4, glucose, C6H12O6, carbon dioxide, CO2 and ammonium phosphate, (NH4) 3PO4, are soluble in water. Which one produces the largest number of dissolved particles per mole of dissolved solute?
A) KCI
B) Na2SO4
C) C6H12O6
D) CO2
E) (NH4) 3PO4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2H2(g) + O2(g) → 2H2O(l)
B) Zn(s) + H2SO4(aq) → ZnSO4(aq) + H2(g)
C) H2O(l) + NH3(g) → NH4+(aq) + OH-(aq)
D) 6FeSO4(aq) + K2Cr2O7(aq) + 7H2SO4(aq) → Cr2(SO4) 3(aq) + 3Fe2(SO4) 3(aq) + K2SO4(aq) + 7H2O(l)
E) Cl2(g) + 2KBr(aq) → Br2(l) + 2KCl(aq)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is most soluble in water?
A) benzene, C6H6
B) potassium nitrate, KNO3
C) carbon tetrachloride, CCl4
D) hexane, C6H14
E) ethane, C2H4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Vinegar is a solution of acetic acid, CH3COOH, dissolved in water. A 5.54-g sample of vinegar was neutralized by 30.10 mL of 0.100 M NaOH. What is the percent by weight of acetic acid in the vinegar?
A) 0.184%
B) 1.63%
C) 3.26%
D) 5.43%
E) 9.23%
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium hydroxide, also known as caustic soda, is used to neutralize acids and to treat cellulose in making of cellophane. Calculate the number of moles of solute in 1.875 L of 1.356 M NaOH solution.
A) 2.543 mol
B) 1.383 mol
C) 0.7232 mol
D) 0.3932 mol
E) 0.001383 mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Automobile batteries use 3.0 M H2SO4 as an electrolyte. How much 1.20 M NaOH will be needed to neutralize 225 mL of battery acid? H2SO4(aq) + 2NaOH(aq) → 2H2O(l) + Na2SO4(aq)
A) 0.045 L
B) 0.28 L
C) 0.56 L
D) 0.90 L
E) 1.1 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many mL of concentrated nitric acid (HNO3, 16.0 M) should be diluted with water in order to make 2.00 L of 2.00 M solution?
A) 32.0 mL
B) 62.5 mL
C) 125 mL
D) 250. mL
E) 500. mL
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 111
Related Exams