B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the net ionic equation for the reaction between lithium hydroxide and hydrobromic acid. LiOH(aq) + HBr(aq) → H2O(l) + LiBr(aq)
A) LiOH(aq) → Li+(aq) + OH-(aq)
B) HBr(aq) → H+(aq) + Br-(aq)
C) H+(aq) + OH-(aq) → H2O(l)
D) Li+(aq) + Br-(aq) → LiBr(aq)
E) Li+(aq) + OH-(aq) + H+(aq) + Br-(aq) → H2O(l) + LiBr(aq)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. Fe(s) + 2Fe3+(aq) → 3Fe2+(aq)
A) Precipitation
B) Acid-base
C) Redox
D) Decomposition
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. 2I-(aq) + Cl2(aq) → I2(aq) + 2Cl-(aq)
A) Combination
B) Displacement
C) Decomposition
D) Precipitation
E) Acid-base
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
Oxidation is associated with an increase in oxidation number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct name and chemical formula for the precipitate that forms when the following reactants are mixed. CoSO4(aq) + (NH4) 3PO4(aq) →
A) cobalt(II) phosphate, Co3(PO4) 2
B) cobalt(III) phosphate, Co3(PO4) 2
C) cobalt(II) phosphate, CoPO4
D) cobalt(III) phosphate, CoPO4
E) ammonium sulfate, (NH4) 2SO4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of H+(aq) ions are present in 750 mL of 0.65 M hydrochloric acid?
A) 1.2 mol
B) 0.98 mol
C) 0.87 mol
D) 0.65 mol
E) 0.49 mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In a redox reaction, the oxidizing agent undergoes loss of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium chlorate is used as an oxidizer in the manufacture of dyes, explosives, and matches. Calculate the mass of solute needed to prepare 1.575 L of 0.00250 M NaClO3 ( 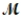 = 106.45 g/mol) .
= 106.45 g/mol) .
A) 419 g
B) 169 g
C) 0.419 g
D) 0.169 g
E) 0.00394 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An acid
A) produces hydroxide ions when dissolved in water.
B) changes the color of phenolphthalein indicator from colorless to pink.
C) donates electrons in an electron transfer reaction.
D) donates protons in a proton transfer reaction.
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the products by completing a balanced equation for the following decomposition reaction. CaCl2(l) 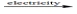 ?
?
A) CaCl2(l) ![]() Ca(l) + 2Cl-(l)
Ca(l) + 2Cl-(l)
B) CaCl2(l) ![]() Ca2+(l) + Cl2(g)
Ca2+(l) + Cl2(g)
C) CaCl2(l) ![]() Ca2+(l) + 2Cl-(l)
Ca2+(l) + 2Cl-(l)
D) CaCl2(l) ![]() Ca(l) + Cl2(g)
Ca(l) + Cl2(g)
E) CaCl2(l) ![]() CaCl(l) + Cl-(l)
CaCl(l) + Cl-(l)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the statements below correctly describes the combustion of glucose, shown below? C6H12O6 + 6O2 → 6CO2 + 6H2O
A) Hydrogen in C6H12O6 is being reduced.
B) Oxygen in O2 is being oxidized.
C) Hydrogen in C6H12O6 is the reducing agent.
D) Oxygen in C6H12O6 is the oxidizing agent.
E) Carbon in C6H12O6 is being oxidized.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solutions will be the best conductor of electrical current?
A) methyl alcohol, CH3OH(aq)
B) glucose, C6H12O6(aq)
C) potassium chloride, KCl(aq)
D) bromine, Br2(aq)
E) ethylene glycol, C2H6O2(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the product(s) for the following reaction. H2SO4(aq) + KOH(aq) →
A) K2SO4(aq) + H2O(l)
B) K2S(aq) + H2O(l)
C) K(s) + H2(g) + SO3(g)
D) KSO4(aq) + H2O(l)
E) No reaction occurs.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
1.0 M aqueous solutions of the following substances are prepared. Which one would you expect to have the lowest electrical conductivity?
A) NaOH
B) CH3CH2OH (ethanol)
C) KBr
D) CH3COOH (acetic acid)
E) HClO4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many milliliters of 1.58 M HCl are needed to react completely with 23.2 g of NaHCO3 ( 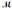 = 84.02 g/mol) ? HCl(aq) + NaHCO3(s) → NaCl(s) + H2O(l) + CO2(g)
= 84.02 g/mol) ? HCl(aq) + NaHCO3(s) → NaCl(s) + H2O(l) + CO2(g)
A) 638 mL
B) 572 mL
C) 536 mL
D) 276 mL
E) 175 mL
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The combustion of an element is always a combination reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ammonia, NH3, produces hydroxide ions in aqueous solution by
A) donating a proton to an acid molecule.
B) donating a proton to a water molecule.
C) donating a hydrogen ion to a water molecule.
D) accepting an electron from an acid molecule.
E) accepting a proton from a water molecule.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A particular reaction may be both a precipitation and an acid-base (neutralization) reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the product(s) for the following reaction. BaO(s) + CO2(g) →
A) Ba(s) + CO3(g)
B) BaCO3(s)
C) BaO(s) + C(s)
D) Ba(s) + CO32-(s)
E) No reaction occurs.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 111
Related Exams