B) False
Correct Answer

verified
Correct Answer
verified
True/False
The entropy of one mole of oxygen gas in a 0.5-L container is less than it would be in a 22.4-L container at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of water is heated at a constant pressure of one atmosphere. Initially, the sample is ice at 260 K, and at the end the sample consists of steam at 400 K. In which of the following 5 K temperature intervals would there be the greatest increase in the entropy of the sample?
A) from 260 K to 265 K
B) from 275 K to 280 K
C) from 360 K to 365 K
D) 370 K to 375 K
E) from 395 K to 400 K
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In order for a process to be spontaneous,
A) the entropy of the system must increase.
B) the entropy of the surroundings must increase.
C) the entropy of the universe must decrease.
D) the entropy of the surroundings must decrease.
E) the entropy change of the surroundings plus the entropy change of the system must be positive.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second law of thermodynamics tells us that
A) the entropy of the universe is constant.
B) entropy is neither created nor destroyed.
C) the universe proceeds towards a state of lower entropy.
D) the universe proceeds towards a state of higher entropy.
E) the universe cannot create entropy.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S for the following reaction? 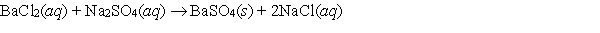
A) ( S 0)
B) ( S < 0)
C) ( S > 0)
D) ( S = H /T)
E) More information is needed to make a reasonable prediction.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a process with S < 0, which one of the following statements is correct?
A) The process will definitely be spontaneous if H < 0.
B) The process will be definitely be spontaneous if H < T S.
C) The process can never be spontaneous.
D) The process will definitely be spontaneous, regardless of H.
E) The process will definitely be spontaneous if Ssurr > 0.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen sulfide decomposes according to the following reaction:  For this reaction at 298 K, S = 78.1 J/K, H = 169.4 kJ, and G = 146.1 kJ. What is the value of G at 900 K?
For this reaction at 298 K, S = 78.1 J/K, H = 169.4 kJ, and G = 146.1 kJ. What is the value of G at 900 K?
A) -69,881 kJ
B) 48.4 kJ
C) 99.1 kJ
D) 240 kJ
E) 441 kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S for the following reaction? 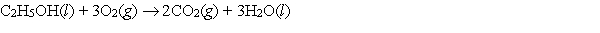
A) ( S 0
B) ( S < 0)
C) ( S > 0)
D) ( S = H /T)
E) More information is needed to make a reasonable prediction.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following changes of state increases the entropy of the system?
A) condensation
B) cooling a gas
C) freezing
D) crystallization
E) sublimation
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the greatest molar entropy at 298 K?
A) CH4(g)
B) H2O(l)
C) NaCl(s)
D) N2O4(g)
E) H2(g)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide.
Correct Answer

verified
Correct Answer
verified
True/False
The term "microstate" refers to the energy state of a single molecule in a system of many molecules.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
For a reaction at equilibrium, Suniv = 0.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
The water-gas shift reaction plays an important role in the production of clean fuel from coal.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S for the following reaction? 
A) ( S 0)
B) ( S < 0)
C) ( S > 0)
D) ( S = H /T)
E) More information is needed to make a reasonable prediction.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the thermodynamic data at 298 K below to determine the Ksp for barium carbonate, BaCO3 at this temperature. 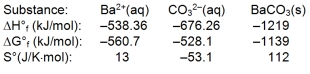
A) 5.86
B) 6.30 * 108
C) 1.59 * 10¯9
D) 5.47 * 10¯21
E) 2.18 * 10¯27
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The higher the pressure of a gas sample, the greater is its entropy.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate G for the reaction of ammonia with fluorine. 
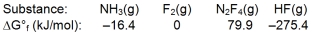
A) 179.1 kJ
B) -179.1 kJ
C) 1539.7 kJ
D) -1539.7 kJ
E) None of these choices is correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes S for the following reaction? 
A) ( S F 0)
B) ( S < 0)
C) ( S > 0)
D) ( S = H /T)
E) More information is needed to make a reasonable prediction.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 85
Related Exams