A) 0.025 mol HCl
B) 0.063 mol HCl
C) 0.082 mol HCl
D) 0.50 mol HCl
E) 1.0 mol HCl
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A CH3COOH/CH3COO¯ buffer can be produced by adding a strong acid to a solution of CH3COO¯ ions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A buffer is prepared by adding 1.00 L of 1.0 M HCl to 750 mL of 1.5 M NaHCOO. What is the pH of this buffer? Ka = 1.7 * 10¯4
A) 2.87
B) 3.72
C) 3.82
D) 3.95
E) 4.66
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following aqueous solutions, when mixed with an equal volume of 0.10 mol L¯1 aqueous NH3, will produce a buffer solution?
A) 0.10 mol L¯1 HCl
B) 0.20 mol L¯1 HCl
C) 0.10 mol L¯1 CH3COOH
D) 0.050 mol L¯1 NaOH
E) 0.20 mol L¯1 NH4Cl
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The solubility of salt MX (solubility product constant Ksp) in water will always be greater than that of salt MX3 (solubility product constant K'sp) provided that Ksp > K'sp.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.25 M HNO3 is titrated with 0.15 M NaOH. What is the pH of the solution after 30.0 mL of NaOH have been added to the acid?
A) 2.00
B) 1.60
C) 1.05
D) 1.00
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a weak acid is titrated with a weak base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0.
D) is determined by the sizes of Ka and Kb.
E) is no longer affected by addition of base.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lab technician Anna Lytic adds 2.20 mol KOH to 1.00 L of 0.5 M Al(NO3) 3. What is the concentration of aluminum ions after the aluminum nitrate has reacted with the potassium hydroxide? Kf = 3.0 * 1033 for Al(OH) 4¯
A) 1.8 * 10¯7 M
B) 9.l * 10¯18 M
C) 1.0 * 10¯31 M
D) 3.3 * 10¯34 M
E) 7.l *10¯36 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicator propyl red has Ka = 3.3 * 10¯6. What would be the approximate pH range over which it would change color?
A) 3.5 to 5.5
B) 4.5 to 6.5
C) 5.5 to 7.5
D) 6.5 to 8.5
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of lead(II) iodide, PbI2, in 0.025 M KI. Ksp = 7.9 *10¯9
A) 4.5 *10¯2 M
B) 2.8 * 10¯2 M
C) 8.9 * 10¯5 M
D) 5.0 * 10¯5 M
E) 1.3 * 10¯5 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum mass of KCl that can be added to 1.0 L of a 0.010 M lead(II) chloride solution without causing any precipitation of lead(II) chloride? Assume that addition of KCl does not affect the solution volume. For lead(II) chloride, Ksp = 1.6 * 10¯5
A) 3.0 g
B) 1.5 g
C) 0.8 g
D) 0.8 g.
E) 0.2 g
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A lab technician adds 0.015 mol of KOH to 1.00 L of 0.0010 M Ca(NO3) 2. Ksp = 6.5 *10¯6 for Ca(OH) 2. Which of the following statements is correct?
A) Calcium hydroxide precipitates until the solution is saturated.
B) The solution is unsaturated and no precipitate forms.
C) The concentration of calcium ions is reduced by the addition of the hydroxide ions.
D) One must know Ksp for calcium nitrate to make meaningful predictions on this system.
E) The presence of KOH will raise the solubility of Ca(NO3) 2.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.50 M H2C6H6O6 (ascorbic acid, a diprotic acid) was titrated with 0.50 M NaOH. The following data were gathered during the titration.  What is Ka2 for ascorbic acid?
What is Ka2 for ascorbic acid?
A) 6.8 *10¯5
B) 6.2 * 10¯6
C) 6.2 * 10¯7
D) 6.2 * 10¯8
E) 2.8 * 10¯12
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A saturated solution of calcium hydroxide, Ca(OH) 2, is in contact with excess solid Ca(OH) 2. Which of the following statements correctly describes what will happen when aqueous HCl (a strong acid) is added to this mixture, and system returns to equilibrium? (For Ca(OH) 2, Ksp = 6.5 * 10¯6)
A) The solubility of Ca(OH) 2 will be unchanged.
B) The OH¯ concentration will decrease and the Ca2+ concentration will increase.
C) The OH¯ concentration will increase and the Ca2+ concentration will decrease.
D) The concentrations of both Ca2+ and OH¯ will increase.
E) The solubility of Ca(OH) 2 will decrease.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An acetic acid buffer containing 0.50 M CH3COOH and 0.50 M CH3COONa has a pH of 4.74. What will the pH be after 0.0020 mol of HCl has been added to 100.0 mL of the buffer?
A) 4.77
B) 4.71
C) 4.68
D) 4.62
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The salts X(NO3) 2 and Y(NO3) 2 (where X+ and Y+ are metal ions) are dissolved in water to give a solution which is 0.1 M in each of them. Which of the answers gives the concentration of chloride ions will precipitate the most YCl2 without precipitating any XCl2? Given Ksp values: XCl2, 2 * 10¯5; YCl2, 1 *10¯10
A) 1 M Cl¯
B) 0.1 M Cl¯
C) 0.01 M Cl¯
D) 0.001 M Cl¯
E) 0.0001 M Cl¯
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Buffer solutions with the component concentrations shown below were prepared. Which of them should have the highest pH?
A) [H2PO4¯] = 0.50 M, [HPO42¯] = 0.50 M
B) [H2PO4¯] = 1.0 M, [HPO42¯] = 1.0 M
C) [H2PO4¯] = 1.0 M, [HPO42¯] = 0.50 M
D) [H2PO4¯] = 0.50 M, [HPO42¯] = 1.0 M
E) [H2PO4¯] = 0.75 M, [HPO42¯] = 1.0 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the dissolution of MnS in water (Ksp = 3.0 * 10¯14) . 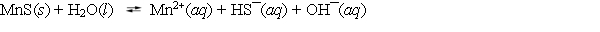 How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
A) The solubility will be unchanged.
B) The solubility will decrease.
C) The solubility will increase.
D) The amount of KOH added must be known before its effect can be predicted.
E) The pKa of H2S is needed before a reliable prediction can be made.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A diprotic acid H2A has Ka1 = 1 *10¯4 and Ka2 = 1 * 10¯8. The corresponding base A2¯ is titrated with aqueous HCl, both solutions being 0.1 mol L¯1. Which one of the following diagrams best represents the titration curve which will be seen?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by adding 100 mL of 0.2 M hydrochloric acid to 100 mL of 0.4 M sodium formate. Is this a buffer solution, and if so, what is its pH?
A) It is a buffer, pH > pKa of formic acid.
B) It is a buffer, pH < pKa of formic acid.
C) It is a buffer, pH = pKa of formic acid.
D) It is a buffer, pH = pKb of sodium formate.
E) Since hydrochloric acid is a strong acid, this is not a buffer.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 115
Related Exams