A) K 2SO 4( aq) + H 2O( l)
B) K 2S( aq) + H 2O( l)
C) K( s) + H 2( g) + SO 3( g)
D) KSO 4( aq) + H 2O( l)
E) No reaction occurs.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of a 0.100 M solution of HCl is required to neutralize a solution prepared by dissolving 10.0 g of KOH in 250 ml of H2O?
A) 0.178 L
B) 0.25 L
C) 0.71 L
D) 1.78 L
E) 178 L
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the precipitate that forms when the following reactants are mixed. Na2CO3(aq) + BaCl2(aq) →
A) Ba 2CO 3
B) BaCO 3
C) NaCl
D) NaCl 2
E) BaO
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What, if any, are the spectator ions when aqueous solutions of HBr and RbOH neutralize each other?
A) H + and OH −
B) H + and Rb +
C) Rb + and Br −
D) Br - and OH −
E) There are no spectator ions in this reaction.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Copper(II) sulfide, CuS, is used in the development of aniline black dye in textile printing. What is the maximum mass of CuS which can be formed when 38.0 mL of 0.500 M CuCl2 are mixed with 42.0 mL of 0.600 M (NH4) 2S? Aqueous ammonium chloride is the other product.
A) 2.41 g
B) 1.82 g
C) 1.21 g
D) 0.909 g
E) 0.044 g
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of ions are released when 0.27 mol of cobalt(II) chloride, CoCl2, is dissolved in water?
A) 0.81 mol
B) 0.54 mol
C) 0.27 mol
D) 0.18 mol
E) 0.090 mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the precipitate that forms when the following reactants are mixed. Mg(CH3COO) 2(aq) + LiOH(aq) →
A) LiCH 3COO
B) Li(CH 3COO) 2
C) MgOH
D) Mg(OH) 2
E) CH 3OH
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.150 M sodium chloride solution is referred to as a physiological saline solution because it has the same concentration of salts as normal human blood. Calculate the mass of solute needed to prepare 275.0 mL of a physiological saline solution.
A) 41.3 g
B) 31.9 g
C) 16.1 g
D) 8.77 g
E) 2.41 g
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous potassium iodate (KIO3) and potassium iodide (KI) react in the presence of dilute hydrochloric acid, as shown below. KIO3(aq) + 5KI(aq) + 6HCl(aq) → 3I2(aq) + 6KCl(aq) + 3H2O(l) What mass of iodine (I2) is formed when 50.0 mL of 0.020 M KIO3 solution reacts with an excess of KI and HCl?
A) 0.13 g I 2
B) 0.25 g I 2
C) 0.38 g I 2
D) 0.76 g I 2
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume, in L, of 10.0 M HCl is needed to make 2.00 L of 2.00 M HCl solution by dilution with water?
A) 0.800 L
B) 0.400 L
C) 0.200 L
D) 0.100 L
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Magnesium carbonate, MgCO3, is insoluble. Identify the spectator ions when aqueous solutions of sodium carbonate and magnesium chloride are combined.
A) Mg 2+ and CO 3 2−
B) Na + and Cl −
C) Mg 2+ and Cl −
D) Na + and CO 3 2−
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
When a solution is diluted with water, the ratio of the initial to final volumes of solution is equal to the ratio of final to initial molarities.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the product(s) for the following reaction. Cl2O7(g) + H2O(l) →
A) HClO 4( aq)
B) H 2ClO 4( aq)
C) H(ClO 4) 2( aq)
D) HCl( aq) + O 2( g)
E) HClO 3( aq)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances, when dissolved in water at equal molar concentrations, will give the solution with the lowest electrical conductivity?
A) CaCl 2
B) HNO 3
C) NH 3
D) C 6H 12O 6 (glucose)
E) CO 2
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
In a redox reaction, the reducing agent undergoes loss of electrons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the classification for the following reaction. CaCl2·H2O(s) 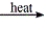 CaCl2(s) + H2O(g)
CaCl2(s) + H2O(g)
A) Combination
B) Decomposition
C) Displacement
D) Acid-base
E) Precipitation
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
In an acid-base (neutralization) reaction the indicator will change color at the end point.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct name and chemical formula for the precipitate that forms when the following reactants are mixed. CuCl2(aq) + Na2CO3(aq) →
A) copper(I) carbonate, Cu 2CO 3
B) copper(II) carbonate, Cu 2CO 3
C) copper(I) carbonate, CuCO 3
D) copper(II) carbonate, CuCO 3
E) sodium chloride, NaCl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a redox reaction?
A) 2Na( s) + 2H 2O( l) → 2NaOH( aq) + H 2( g)
B) H 2( g) + Cl 2( g) → 2HCl( g)
C) 2H 2O 2( aq) → 2H 2O( l) + O 2( g)
D) Fe 2O 3( s) + 3H 2SO 4( aq) → Fe 2(SO 4) 3( aq) + 3H 2O( l)
E) 2KMnO 4( aq) + 10FeSO 4( aq) + 8H 2SO 4( aq) → K 2SO 4( aq) + 2MnSO 4( aq) + 5Fe 2(SO 4) 3( aq) + 8H 2O( l)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a strong base?
A) NH 3
B) Ca(OH) 2
C) Al(OH) 3
D) B(OH) 3
E) CH 3OH
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 111
Related Exams