A) HOC 6H 4OCOOH, K a = 1.0 × 10 −3
B) C 6H 4(COOH) 2, K a = 2.9 × 10 −4
C) CH 3COOH, K a = 1.8 × 10 �−5
D) C 5H 5O 5COOH, K a = 4.0 × 10 −6
E) HBrO, K a = 2.3 × 10 −9
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Equal volumes of the following pairs of solutions are mixed. Which pair will produce a buffer solution?
A) 0.10 mol L −1 HCl and 0.05 mol L −1 NaOH
B) 0.10 mol L −1 HCl and 0.15 mol L −1 NH 3
C) 0.10 mol L −1 HCl and 0.05 mol L −1 NH 3
D) 0.10 mol L −1 HCl and 0.20 mol L −1 CH 3COOH
E) 0.10 mol L −1 HCl and 0.20 mol L −1 NaCl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, of the following aqueous mixtures would be a buffer system?
A) CH 3COOH, NaH 2PO 4
B) H 2CO 3, HCO 3 −
C) H 2PO 4 − , HCO 3 −
D) HSO 4 − , HSO 3 −
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A change in pH will significantly affect the solubility of which, if any, of the following compounds?
A) BaF 2
B) CuCl
C) CuBr
D) AgI
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a buffer that consists of 0.20 M NaH2PO4 and 0.40 M Na2HPO4? For NaH2PO4, Ka = 6.2 × 10−8
A) 6.51
B) 6.91
C) 7.51
D) 7.90
E) 8.13
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of silver chloride __________ when dilute nitric acid is added to it.
A) increases
B) decreases
C) does not change
D) first increases, then decreases
E) first decreases, then increases
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Citric acid has an acid dissociation constant of 8.4 × 10−4. It would be most effective for preparation of a buffer with a pH of
A) 2.
B) 3.
C) 4.
D) 5.
E) 6.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of NaF must be added to 50.0 mL of a 0.500 M HF solution to achieve a pH of 3.25? For HF, Ka = 7.2 × 10−4.
A) 1.3 g
B) 0.69 g
C) 6.9 g
D) 23 g
E) 1.5 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the ion product expression for calcium phosphate, Ca3(PO4) 2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At the equivalence point in an acid-base titration
A) the [H 3O +] equals the K a of the acid.
B) the [H 3O +] equals the K a of the indicator.
C) the amounts of acid and base which have been combined are in their stoichiometric ratio.
D) the pH is 7.0.
E) the pH has reached a maximum.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming that the total volume does not change after 0.200 g of KCl is added to 1.0 L of a saturated aqueous solution of AgCl, calculate the number of moles of Ag+ ion in the solution after equilibrium has been reestablished. For AgCl, Ksp = 1.8 × 10−10.
A) 1.8 × 10 −10 mol Ag +
B) 9.0 × 10 −10 mol Ag +
C) 9.0 × 10 −9 mol Ag +
D) 6.7 × 10 −8 mol Ag +
E) 1.3 × 10 −5 mol Ag +
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.30 M HClO was titrated with 0.30 M NaOH. The following data were collected during the titration.  What is the Ka for HClO?
What is the Ka for HClO?
A) 1.1 × 10 −7
B) 3.5 × 10 −8
C) 1.2 × 10 −8
D) 4.9 × 10 −11
E) None of these choices are correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The equivalence point in a titration is defined as the point when the indicator changes color.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of magnesium sulfate, MgSO4, when placed into a 0.10 M MgCl2 solution. Ksp = 5.9 × 10−3
A) 4.2 × 10 −2 M
B) 5.9 × 10 −2 M
C) 7.7 × 10 −2 M
D) 3.5 × 10 −5 M
E) 3.5 × 10 −6 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Buffer solutions with the component concentrations shown below were prepared. Which of them should have the highest pH?
A) [H 2PO 4 −] = 0.50 M, [HPO 4 2−] = 0.50 M
B) [H 2PO 4 −] = 1.0 M, [HPO 4 2−] = 1.0 M
C) [H2PO4−] = 1.0 M, [HPO42−] = 0.50 M
D) [H2PO4−] = 0.50 M, [HPO42−] = 1.0 M
E) [H 2PO 4 −] = 0.75 M, [HPO 4 2−] = 1.0 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of silver chromate, Ag2CrO4, in 0.005 M Na2CrO4. Ksp = 2.6 × 10−12
A) 1.4 × 10−4 M
B) 3.4 × 10 −5 M
C) 1.1 × 10 −5 M
D) 1.6 × 10 −6 M
E) < 1.0 × 10 −6 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When 20.0 mL of 0.15 M hydrochloric acid is mixed with 20.0 mL of 0.10 M sodium hydroxide, the pH of the resulting solution is
A) 0.00.
B) 12.40.
C) 1.60.
D) 0.82.
E) 7.00.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve that will be obtained in the titration of a weak acid (0.10 mol L−1) with a strong base of the same concentration? 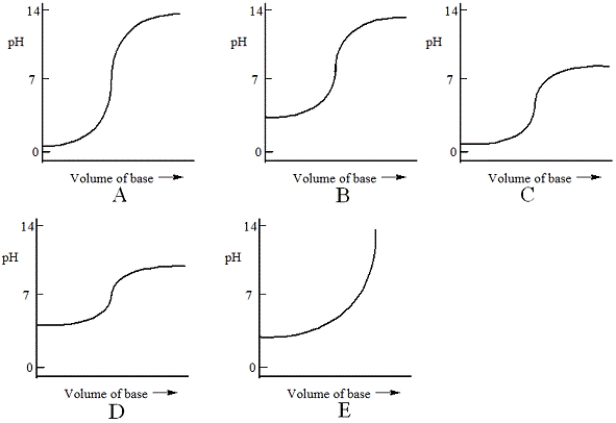
A) A
B) B
C) C
D) D
E) E
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of silver oxalate, Ag2C2O4, in pure water. Ksp = 1.0 × 10−11
A) 1.4 × 10 −4 M
B) 8.2 × 10 −5 M
C) 5.4 × 10 −5 M
D) 3.2 × 10 −6 M
E) 2.5 �× 10 −12 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of silver phosphate, Ag3PO4, in pure water. Ksp = 2.6 × 10−18
A) 4.0 × 10 −5 M
B) 1.8 × 10 −5 M
C) 4.0 × 10 −6 M
D) 1.5 × 10 −5 M
E) < 1.0 × 10 −5 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 114
Related Exams