A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the minimum concentration of Mg2+ that must be added to 0.10 M NaF in order to initiate a precipitate of magnesium fluoride. [Ksp(MgF2) = 6.9 × 10-9]
A) 1.4 × 107 M
B) 6.9 × 10-9 M
C) 6.9 × 10-8 M
D) 1.7 × 10-7 M
E) 6.9 × 10-7 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of the sparingly soluble salt MX at equilibrium in aqueous solution. (Each circle represents 1.0 ×10-3 mol of atoms, and the volume of the box is 1.0 L. Solvent water molecules are omitted for clarity.) 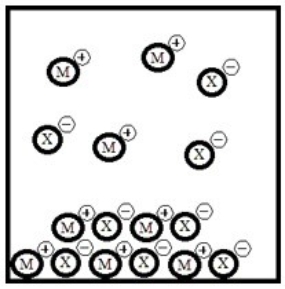 What is Ksp for MX?
What is Ksp for MX?
A) 1.5 ×10-5
B) 6.0 ×10-6
C) 3.6 ×10-1
D) 1.8 ×10-3
E) 9.0 ×10-6
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A CH3COOH/CH3COO- buffer can be produced by adding a strong acid to a solution of CH3COO- ions.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
The ________ is the solution that is added from the buret during a titration.
Correct Answer

verified
Correct Answer
verified
True/False
Increasing the concentrations of the components of a buffer solution will increase the buffer capacity.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a weak acid is titrated with a strong base, the pH at the equivalence point
A) is greater than 7.0.
B) is equal to 7.0.
C) is less than 7.0.
D) is equal to the pKa of the acid.
E) is equal to the pKb of the conjugate base.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You have 500.0 mL of a buffer solution containing 0.20 M acetic acid (CH3COOH) and 0.30 M sodium acetate (CH3COONa) . What will the pH of this solution be after the addition of 20.0 mL of 1.00 M NaOH solution? [Ka(CH3COOH) = 1.8 × 10-5]
A) 4.41
B) 4.74
C) 4.56
D) 4.92
E) 5.07
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution that consists of 0.50 M H2C6H6O6 (ascorbic acid) and 0.75 M NaHC6H6O6 (sodium ascorbate) ? [Ka(H2C6H6O6) = 6.8 × 10-5]
A) 3.76
B) 3.99
C) 4.34
D) 4.57
E) 5.66
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 50.00-mL solution of 0.10 M HNO2 is titrated with a 0.10 M KOH solution. After 25.00 mL of the KOH solution is added, what is the pH in the titration flask? [Ka(HNO2) = 4.5 × 10-4]
A) 2.17
B) 3.35
C) 2.41
D) 1.48
E) 7.00
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose 0.015 mol of KOH is added to 1.00 L of 0.0010 M Ca(NO3) 2. Which statement is correct? [Ksp(Ca(OH) 2) = 6.5 × 10-6]
A) Calcium hydroxide precipitates until the solution is saturated.
B) The solution is unsaturated and no precipitate forms.
C) The concentration of calcium ions is reduced by the addition of the hydroxide ions.
D) Potassium nitrate precipitates until the solution is saturated.
E) The presence of KOH will raise the solubility of Ca(NO3) 2.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a solution that consists of 1.5 M NH3 and 2.5 M NH4Cl? Kb = 1.8 × 10-5
A) 1.1 × 10-5 M
B) 3.0 × 10-6 M
C) 3.3 × 10-9 M
D) 5.6 × 10-10 M
E) 9.3 × 10-10 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose 50.0 mL of a 0.100 M solution of the weak acid HA is titrated with a 0.100 M solution of NaOH. Which diagram best represents the equilibrium composition of the solution once 25.0 mL of titrant have been added? (Solvent water molecules are omitted for clarity.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH at the equivalence point in the titration of 100 mL of 0.10 M HCl with 0.10 M NaOH?
A) 1.0
B) 6.0
C) 7.0
D) 8.0
E) 13.0
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the effect on equilibrium when sodium acetate is added to a solution of acetic acid? CH3COOH(aq)  H+(aq) + CH3COO-(aq)
H+(aq) + CH3COO-(aq)
A) There is no change in the equilibrium.
B) The equilibrium shifts to the right.
C) More information is needed to answer the question.
D) The equilibrium shifts to the left.
E) The pH decreases.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a buffer solution prepared by dissolving 0.20 mole of sodium cyanate (NaCNO) and 1.0 mole of cyanic acid (HCNO) in enough water to make 1.0 liter of solution. [Ka(HCNO) = 2.0 × 10-4]
A) 0.00
B) 3.00
C) 3.70
D) 4.40
E) 5.00
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
For the 1:1 ratio combination of a monoprotic strong acid and a monoprotic strong base, the ________ is where equal molar amounts of the strong acid and strong base have been combined.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicator propyl red has Ka = 3.3 × 10-6. What would be the approximate pH range over which it would change color?
A) 3.5 - 5.5
B) 4.5 - 6.5
C) 5.5 - 7.5
D) 6.5 - 8.5
E) 7.5 - 9.5
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the Henderson-Hasselbach equation?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the dissolution of MnS in water. [Ksp(MnS) =3.0 × 10-14] MnS(s) + H2O(l) ![Consider the dissolution of MnS in water. [K<sub>sp</sub>(MnS) =3.0 × 10<sup>-14</sup>] MnS(s) + H<sub>2</sub>O(l) Mn<sup>2+</sup>(aq) + HS<sup>-</sup>(aq) + OH<sup>-</sup>(aq) How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system? A) The solubility will be unchanged. B) The solubility will decrease. C) The solubility will increase. D) The amount of KOH added must be known before its effect can be predicted. E) The pK<sub>a</sub> of H<sub>2</sub>S is needed before a reliable prediction can be made.](https://d2lvgg3v3hfg70.cloudfront.net/TB8482/11eb6c5c_3a64_3a31_8d9d_d73b5c050f32_TB8482_11.jpg) Mn2+(aq) + HS-(aq) + OH-(aq)
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
Mn2+(aq) + HS-(aq) + OH-(aq)
How is the solubility of manganese(II) sulfide affected by the addition of aqueous potassium hydroxide to the system?
A) The solubility will be unchanged.
B) The solubility will decrease.
C) The solubility will increase.
D) The amount of KOH added must be known before its effect can be predicted.
E) The pKa of H2S is needed before a reliable prediction can be made.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 133
Related Exams