A) 171 J
B) 393 J
C) 504 J
D) 615 J
E) 837 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The work done on the surroundings by the expansion of a gas is w = -PΔV.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the standard enthalpy change for the decomposition of one mole of SO3? 2SO2 + O2→ 2SO3 ΔH°rxn = -198 kJ/mol
A) 198 kJ/mol
B) -99.0 kJ/mol
C) 99.0 kJ/mol
D) 396 kJ/mol
E) -396 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system absorbs 21.6 kJ of heat while performing 6.9 kJ of work on the surroundings. If the initial internal energy, U, is 61.2 kJ, what is the final value of U?
A) 32.7 kJ
B) 46.5 kJ
C) 61.2 kJ
D) 75.9 kJ
E) 89.7 kJ
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat (capacity) is
A) the amount of energy needed to increase the temperature of 1 g of a substance by 1°C.
B) the amount of energy needed to increase the temperature of 1 mol of a substance by 1°C.
C) the amount of energy required to melt 1 g of substance.
D) the amount of substance that is heated by 1°C.
E) the temperature increase, in K, associated with heating 1 g of a substance for 1 minute.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system that does no work but which receives heat from the surroundings has
A) q < 0, ΔU > 0.
B) q > 0, ΔU < 0.
C) q = ΔU.
D) q = -ΔU.
E) w = ΔU.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An important step in the synthesis of nitric acid is the conversion of ammonia to nitric oxide according to the following balanced chemical equation. What is ΔH°rxn for this reaction? 4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(g) 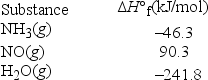
A) -1274.8 kJ/mol
B) -904.4 kJ/mol
C) -240.2 kJ/mol
D) -197.8kJ
E) 197.8 kJ/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A Snickers® candy bar contains 280 Calories, of which the fat content accounts for 120 Calories. What is the energy of the fat content, in kJ? (1 cal = 4.184 J)
A) 5.0 × 10-1 kJ
B) 1.2 kJ
C) 5.0 × 102 kJ
D) 1.2 × 103 kJ
E) 1.6 × 103 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The sign of q when heat is absorbed by the system is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A student used a bomb calorimeter to determine the heat of combustion of an unknown compound. If the student ignored the heat capacity of the bomb calorimeter, the heat capacity of the unknown compound would be ________ the accepted value.
A) greater than (i.e., less negative)
B) less than (i.e., more negative)
C) equal to
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much heat is required to raise the temperature of 12.0 g of water from 15.4°C to 93.0°C? The specific heat of water is 4.184 J/g·°C.
A) 223 J
B) 773 J
C) 503 J
D) 4.67 ×103 J
E) 3.90 ×103 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethylene glycol, used as a coolant in automotive engines, has a specific heat capacity of 2.42 J/g°C. Calculate q when 3.65 kg of ethylene glycol is cooled from 132°C to 85°C.
A) -1900 kJ
B) -420 kJ
C) -99 kJ
D) -0.42 kJ
E) -4.2 × 10-6 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which type of system may transfer energy, but not mass, to the surroundings?
A) mass transfer system
B) isolated system
C) closed system
D) nonenergy system
E) open system
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system that does no work but which transfers heat to the surroundings has
A) q < 0, ΔU > 0.
B) q < 0, ΔU < 0.
C) q > 0, ΔU > 0.
D) q > 0, ΔU < 0.
E) q < 0, ΔU = 0.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Styrene, C8H8, is one of the substances used in the production of synthetic rubber. When styrene burns in oxygen to form carbon dioxide and liquid water under standard-state conditions at 25°C, 42.15 kJ are released per gram of styrene. Find the standard enthalpy of formation of styrene at 25°C. ΔH°f(CO2(g) ) = -393.5 kJ/mol, ΔH°f(H2O(l) ) = -285.8 kJ/mol
A) -4390 kJ/mol
B) -1044 kJ/mol
C) -8681 kJ/mol
D) +99 kJ/mol
E) +637 kJ/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In an endothermic process, heat is absorbed by the system.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose a sample of gas undergoes the following change of state. 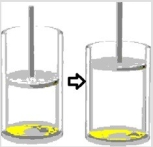 Which statement(s) is/are true concerning this change of state?
Which statement(s) is/are true concerning this change of state? 
A) I only
B) II only
C) II and III
D) II and IV
E) I and III
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat of solution of ammonium chloride is 15.2 kJ/mol. If a 6.134-g sample of NH4Cl is added to 65.0 mL of water in a calorimeter at 24.5°C, what is the final temperature of the solution? The specific heat of water is 4.18 J/g·°C and the heat capacity of the calorimeter is 365 J/°C.
A) 27.1°C
B) 18.6°C
C) 19.7°C
D) 21.8°C
E) 30.4°C
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An exothermic reaction causes the surroundings to
A) warm up.
B) become acidic.
C) expand.
D) decrease its temperature.
E) release CO2.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond enthalpy of the Br-Cl bond is equal to ΔH°rxn for the following reaction. BrCl(g) → Br(g) + Cl(g)
Using the following data, what is the bond enthalpy of the Br-Cl bond? 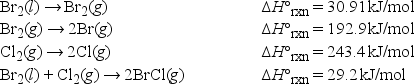
A) 219.0 kJ/mol
B) 203.5 kJ/mol
C) 14.6 kJ/mol
D) 438.0 kJ/mol
E) 407.0 kJ/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 141
Related Exams