A) greatest on Day One.
B) greatest on Day Two.
C) the same on both days.
D) zero on both days.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At the dice factory, sets of novelty dice are created where a side with 7 dots replaces the single dot side.The sides then range from 2 to 7 instead of the usually 1 to 6.Using such dice for a game of craps, what will be the most probable roll?
A) 7, this change in dots results in the same most probable value.
B) 7.5
C) 8
D) 9
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pound of body fat has an energy content of about 4 100 kcal.If a 1 400-kg automobile had an equivalent amount of translational kinetic energy, how fast would it be moving? (0.447 m/s = 1 mph, 1 kcal = 4 186 J)
A) 3.1 mph
B) 14 mph
C) 75 mph
D) 350 mph
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The volume of an ideal gas changes from 0.40 to 0.55 m3 although its pressure remains constant at 50 000 Pa.What work is done on the system by its environment?
A) -7 500 J
B) -200 000 J
C) 7 500 J
D) 200 000 J
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 2.0-mol ideal gas system is maintained at a constant volume of 4.0 L.If 100 J of heat is added, what is the work done on the system?
A) zero
B) 5.0 J
C) -6.7 J
D) 20 J
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following choices best corresponds to what is required by the second law of thermodynamics for any process taking place in an isolated system?
A) entropy decreases
B) entropy remains constant
C) entropy increases
D) entropy equals work done on the system
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an isovolumetric process where the pressure increases, are the heat absorbed, work done by the system, and the change in internal energy of the system positive, negative, or zero?
A) Q is +, W is +, and DU is +.
B) Q is +, W is -, and DU is 0.
C) Q is +, W is 0, and DU is +.
D) Q is -, W is 0, and DU is -.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 2.00-kg block of ice is at STP (0 C, 1 atm) while it melts completely to water.What is its change in entropy? (For ice, Lf = 3.34 * 105 J/kg)
A) zero
B) 584 J/K
C) 1 220 J/K
D) 2 450 J/K
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Entropy is a measure of the ____ of a system.
A) disorder
B) temperature
C) heat
D) internal energy
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Area on a P-V diagram has units associated with:
A) energy.
B) momentum.
C) temperature.
D) change in temperature.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal gas at pressure, volume, and temperature, P0, V0, and T0, respectively, is heated to point A, allowed to expand to point B also at A's temperature 2T0, and then returned to the original condition.The internal energy decreases by 3P0V0/2 going from point B to point T0.In going around this cycle once, which quantity equals zero? 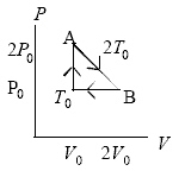
A) the net change in internal energy of the gas
B) the net work done by the gas
C) the net heat added to the gas
D) All three are zero.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cylinder containing an ideal gas has a volume of 2.0 m3 and a pressure of 1.0 * 105 Pa at a temperature of 300 K.The cylinder is placed against a metal block that is maintained at 900 K and the gas expands as the pressure remains constant until the temperature of the gas reaches 900 K.The change in internal energy of the gas is 6.0 * 105 J.Find the change in entropy of the block associated with the heat transfer to the gas.
A) 0
B) +670 J/K
C) -440 J/K
D) -1 100 J/K
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The adiabatic index of a gas is given by which of the following?
A) CP /CV
B) CV /CP
C) CP - CV
D) CP + CV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During an isobaric process which one of the following does not change?
A) volume
B) temperature
C) internal energy
D) pressure
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 4-mol ideal gas system undergoes an adiabatic process where it expands and does 20 J of work on its environment.How much heat is received by the system?
A) -20 J
B) zero
C) +5 J
D) +20 J
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Carnot cycle consists of a combination of ____ and ____ processes.
A) isobaric, isovolumetric
B) isovolumetric, adiabatic
C) isobaric, isothermal
D) adiabatic, isothermal
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a heat engine has an efficiency of 30% and its power output is 600 W, what is the rate of heat input from the combustion phase?
A) 1 800 W
B) 2 400 W
C) 2 000 W
D) 3 000 W
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A heat engine receives 6 000 J of heat from its combustion process and loses 4 000 J through the exhaust and friction.What is its efficiency?
A) 33%
B) 40%
C) 67%
D) 73%
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Heat is applied to an ice-water mixture to melt some of the ice.In this process:
A) work is done by the ice-water mixture.
B) the temperature increases.
C) the internal energy increases.
D) all of the above are correct.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the first law of thermodynamics, for any process that may occur within an isolated system, which of the following choices applies?
A) entropy remains constant
B) entropy increases
C) entropy decreases
D) None of the above choices apply.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 70
Related Exams