B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy associated with vibrations within molecules is called _____.
A) activation energy
B) surface energy
C) internal energy
D) elastic potential energy
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following energy diagram, which letter represents the catalyzed activation energy? 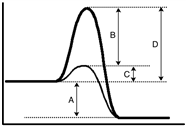
A) A
B) B
C) C
D) D
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the statements is correct when referring to the following reaction? H2(g) + I2(g)  2HI(g)
2HI(g)
A) This reaction is reversible because the amount of heat given off by the forward reaction is the same as the heat given off by the reverse reaction.
B) The physical state of the reaction is found by using ![]() .
.
C) Most of the matter is on the right side of the equation.
D) There isn't a correct response provided.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be the equilibrium expression for the system given below? N2(g) + 3F2(g)  2NF3(g)
2NF3(g)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The reaction below does not progress at room temperature, but does at 1100 ° C. Therefore, this reaction is spontaneous at 1100 ° C.
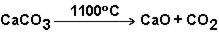
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, which of the given changes will increase the value of the equilibrium constant? AB + CD  AD + BC + heat
AD + BC + heat
A) decreasing the temperature
B) adding AB to the reaction
C) removing CD from the reaction
D) All of these changes will increase K .
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based upon the energy diagram given below, which letter represents the enthalpy for the reaction? 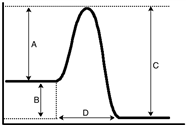
A) A
B) B
C) C
D) D
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Chemical reactions always occur once the molecules of two substances collide.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement applies to a reaction at equilibrium?
A) The energy of activation has been supplied to bring the reaction to equilibrium.
B) The state of the equilibrium can be expressed as a ratio.
C) There are equal amounts of matter on either side of the reaction.
D) All of the responses are correct.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
A reaction rate can be described using any unit of time (seconds, hours, etc.).
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Butane, C4H10, burning in air, is an example of a substance combining with oxygen to produce CO2 and H2O. If pure oxygen were to be supplied, the rate of combustion would increase significantly. Then there is more energy given off when 10 g of butane burns in pure oxygen, rather than in air.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an endergonic process?
A) exothermic reactions
B) endothermic reactions
C) equilibrium reactions
D) none of them
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is NOT true regarding a chemical reaction?
A) All matter is considered to be composed of tiny particles.
B) All particles contain potential energy, measured by constant motion.
C) Particle speed always increases with rising temperature.
D) Colliding particles will transfer energy with no loss in system energy.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ice has a(n) ____ entropy than that of liquid water.
A) lower
B) equal
C) higher
D) can not tell
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The device used to measure heat produced or used by a chemical reaction is called a(n) _____ .
A) thermometer
B) calorimeter
C) activator
D) joulery
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is incorrect with reference to Le Châtelier's principle?
A) In a reaction of the type, Q + 2R ![]() 3T, the addition of one mole of Q results in the tripling of T.
3T, the addition of one mole of Q results in the tripling of T.
B) If the reaction to the right is exothermic, the reaction to the left must be endothermic.
C) If the reaction rate of the forward reaction is known, then the reaction rate of the reverse reaction is also known.
D) The reaction to the left is neither exothermic, nor is the reaction rate of the reverse reaction known
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
For the reaction NO2 (g) + N2O(g)  3 NO(g)the equilibrium constant expression is:
3 NO(g)the equilibrium constant expression is:
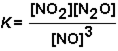
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In an exergonic process, the system _____ .
A) gains energy
B) loses energy
C) either gains or loses energy
D) no energy change at all
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A process that gains or accepts energy as it takes place is a(n) _____ .
A) exergonic process
B) spontaneous process
C) endergonic process
D) all of these
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 92
Related Exams