A) NH4Cl and NH3
B) HNO3 and NaNO3
C) HF and KF
D) C6H5COOH (Benzoic acid) and C6H5COO - Na+
E) CH3COOH and CH3COO - Na+
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the two buffer solutions prepared below: Solution A: [HA] = 0.15 M and [A - ] = 0.25 M Solution B: [HA] = 1.50 M and [A - ] = 2.50 M Which statement below is true regarding the pH and buffering capacity of these two solutions?
A) Solution A will have a greater pH value and a smaller buffering capacity.
B) Solution B will have a greater pH value and a smaller buffering capacity.
C) Solution B will have a greater pH value and a larger buffering capacity.
D) The pH of both solutions will be identical but Solution A will have a larger buffering capacity.
E) The pH of both solutions will be identical but Solution B will have a larger buffering capacity.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbonic acid is a diprotic acid with K al = 4.3×10 - 7 and K a2 = 5.6×10 - 11. Estimate the pH of a solution of sodium hydrogen carbonate.
A) pH = 2
B) pH = 4
C) pH = 6
D) pH = 8
E) pH = 10
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following salts, each having very limited solubility in water, would dissolve to a greater extent upon acidifying the solution? I. Ag2S II. Mg3(PO4) 2 III. AgBr
A) I only
B) II only
C) I and II
D) All of these
E) None of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following titrations would produce a pH equal to 7.0 at the equivalence point? I. Titrating NH3 with standard HCl (delivered from the burette) II. Titrating HCl with standard NaOH (delivered from the burette) III. Titrating CH3COOH with standard NaOH (delivered from the burette)
A) I only
B) II only
C) III only
D) II and III
E) All of these
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following general combinations of substances form an acid-base buffer when added to water?
A) a strong acid and the sodium salt of its conjugate base
B) a strong base and the chloride salt of its conjugate acid
C) a weak acid and the sodium salt of its conjugate base
D) a weak acid
E) a weak base and a strong base
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a buffer prepared by dissolving 0.10 mol of NH4Cl in 1.00 L of 0.15 M NH3. K b = 1.8×10 - 5 for NH3.
A) 9.43
B) 4.56
C) 11.34
D) 2.72
E) 4.74
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the volume of 0.0175 M Ba(OH) 2 required to neutralize 10.0 mL of 0.0300 M HCl?
A) 10.0 mL
B) 34.2 mL
C) 17.1 mL
D) 8.6 mL
E) 4.3 mL
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of 0.100 mole of acetic acid ( K a = 1.8×10 - 5) in 50.0 mL of solution is titrated with standard 0.100 M NaOH. What is the pH in the titration flask after addition of 25.0 mL of the base?
A) 2.88
B) 3.18
C) 2.60
D) 4.74
E) 11.40
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the pH of a solution formed when 1.5 L oF4.5×10 - 3 M HCl is mixed witH₂ .0 L of 3.0×10 - 3 M HNO3.
A) 3.67
B) 3.74
C) 2.25
D) 3.16
E) 2.44
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Exhibit 16-1 Consider the equilibrium reaction below to answer the following question(s) . HNO₂ + H₂ O ![Exhibit 16-1 Consider the equilibrium reaction below to answer the following question(s) . HNO₂ + H₂ O H₃O + + NO₂ 1 - K a (HNO₂ ) = 4.5 10 - 4 -Refer to Exhibit 16-1. What is the concentration of nitrite ion, NO₂ - , present at equilibrium if 0.100 moles of HCl are added to 0.200 moles of HNO₂ to form one-liter of solution? A) [NO<sub>2</sub><sup> - </sup>] = 9.00×10<sup> - 4</sup> M B) [NO<sub>2</sub><sup> - </sup>] = 2.12×10<sup> - 3</sup> M C) [NO<sub>2</sub><sup> - </sup>] = 9.49×10<sup> - 3</sup> M D) [NO<sub>2</sub><sup> - </sup>] = 0.100 M E) [NO<sub>2</sub><sup> - </sup>] = 0.200 M](https://d2lvgg3v3hfg70.cloudfront.net/TBX8714/11ebff50_c8c3_2262_8da6_116167ac28d4_TBX8714_11.jpg) H₃O + + NO₂ 1 - K a (HNO₂ ) = 4.5 10 - 4
-Refer to Exhibit 16-1. What is the concentration of nitrite ion, NO₂ - , present at equilibrium if 0.100 moles of HCl are added to 0.200 moles of HNO₂ to form one-liter of solution?
H₃O + + NO₂ 1 - K a (HNO₂ ) = 4.5 10 - 4
-Refer to Exhibit 16-1. What is the concentration of nitrite ion, NO₂ - , present at equilibrium if 0.100 moles of HCl are added to 0.200 moles of HNO₂ to form one-liter of solution?
A) [NO2 - ] = 9.00×10 - 4 M
B) [NO2 - ] = 2.12×10 - 3 M
C) [NO2 - ] = 9.49×10 - 3 M
D) [NO2 - ] = 0.100 M
E) [NO2 - ] = 0.200 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Oxalic acid is diprotic with p K al = 1.23 and p K a2 = 4.19. What is p K b for the hydrogen oxalate ion?
A) 1.23
B) 4.19
C) 7.00
D) 9.81
E) 12.77
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of acetic acid (approximately 0.001 M, K a = 1.8×10 - 5) is titrated with 0.00993 M NaOH. What indicator should be used?
A) thymol blue (p K In = 1.65)
B) methyl orange (p K In = 3.46)
C) methyl red (p K In = 5.00)
D) bromthymol blue (p K In = 7.30)
E) phenolphthalein (p K In = 8.7)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.10 M solution of hydrazine (N2H4, K b = 1.7×10 - 6) containing an unknown concentration of hydrazine hydrochloride (N2H5Cl) has a pH of 7.50. What is the concentration of N2H5Cl in the solution?
A) 1.26 M
B) 0.54 M
C) 5.8 M
D) 0.10 M
E) 0.72 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A buffer was prepared such that the pH was 5.20. If the solution contains 0.400 moles of acetic acid (CH3COOH) per liter, what is the concentration of the acetate ion (CH3COO - ) in this solution? The p K a for acetic acid is 4.74.
A) 0.40 M
B) 0.14 M
C) 4.2×10 - 3 M
D) 6.3×10 - 6 M
E) 1.1 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the titration of 25.0 mL of a solution of a weak acid HX with 0.250 M NaOH, the pH = 4.50 after the addition of 5.0 mL of the NaOH solution. The equivalence point occurs when 30.4 mL of the NaOH solution has been added. What is the ionization constant ( K a) of HX?
A) 1.4×10 - 5
B) 3.2×10 - 5
C) 6.2�×10 - 6
D) 3.2×10 - 10
E) 7.2×10 - 10
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the titration of a weak acid with a strong base . Which statement below is true regarding the equivalence point in this titration?
A) It occurs at a pH = 7.0 because the acid has been neutralized.
B) It occurs at a pH less than 7.0 because an acidic salt is formed.
C) It occurs at a pH less than 7.0 because a basic salt is formed.
D) It occurs at a pH greater than 7.0 because an acidic salt is formed.
E) It occurs at a pH greater than 7.0 because a basic salt is formed.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Solutions are made by combining equal amounts of the following. Which is a buffer?
A) 0.1 M HCl + 0.1 M Cl -
B) 0.1 M HF + 0.1 M Na+
C) 0.1 M HF + 0.1 M NaOH
D) 0.1 M NaF + 0.05 M HF
E) 0.1 M NaF + 0.05 M Na+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a buffer solution containing a weak acid, HX, and a salt of the weak acid's conjugate base, NaX. Under what conditions is the pH greater than the p K a of the weak acid?
A) When the p K a equals 7.0.
B) When [HX] > [NaX].
C) When [HX] < [NaX].
D) When [HX] = [NaX].
E) When there is no NaX present.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of 0.200 M H₂ SO₄is required to neutralize 50.0 mL of 0.100 M NaOH?
Assume the chemical equation is:
H₂ SO₄(aq) + 2 NaOH (aq) Na2 SO₄(aq) + 2 H₂ O ( 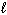 )
)
A) 50.0 mL
B) 25.0 mL
C) 100 mL
D) 12.5 mL
E) 6.25 mL
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 98
Related Exams