A) For an organic compound with one functional group that contains an O or N atom, the compound is water soluble only if it has 35 carbons.
B) The non-polar part of a molecule that is not attracted to water is said to be hydrophilic.
C) The polar part of a molecule that can that can bond hydrogen to water is said to be hydrophobic.
D) The non-polar part of a molecule that is not attracted to water is said to be hydrophobic.
F) C) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Consider the molecule atenolol (a ? blocker used to treat hypertension) . Which of the following lists the correct functional groups present in atenolol? 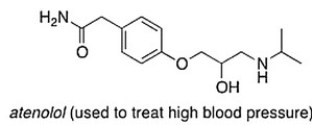
A) Secondary alcohol, amide, secondary amine, aromatic, ester
B) Secondary alcohol, amide, primary amine, aromatic, ether
C) Secondary alcohol, amide, secondary amine, aromatic, ether
D) Primary alcohol, amide, primary amine, aromatic, ether
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the intermolecular forces present in the following molecule: 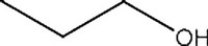
A) Dipole-dipole interactions
B) More than one of these answer choices is correct.
C) Van der Waals
D) Hydrogen bonding
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 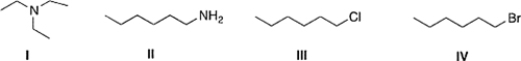
A) IV
B) I
C) III
D) II
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements best describes the relationship between the surface area of a molecule and the strength of the intermolecular forces?
A) The smaller the surface area, the stronger the intermolecular forces.
B) The larger the surface area, the stronger the intermolecular forces.
C) The larger the surface area, the weaker the intermolecular forces.
D) There is no relationship between surface area and intermolecular forces.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is one of the families of biomolecules?
A) Simple sugars
B) Amino acids
C) All of these are correct.
D) Lipids
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 
A) IV
B) II
C) III
D) I
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 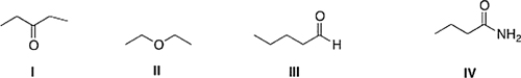
A) II
B) III
C) I
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which C=O functional group is present in the following molecule? 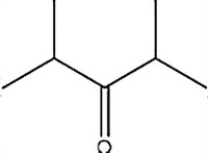
A) Aldehyde
B) Ketone
C) Ester
D) Carboxylic acid
F) B) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point? 
A) II
B) III
C) IV
D) I
F) None of the above
Correct Answer

verified
C
Correct Answer
verified
Showing 1 - 10 of 10
Related Exams