A) It reacts with base to form water.
B) Its density is 1.84 g/mL.
C) It tastes sour.
D) It is a white-colored solid.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate whether each of the following statements represents a chemical change, a physical change, or no change occurred. -A chunk of cheddar cheese is grated. ____________________
Correct Answer

verified
Correct Answer
verified
Essay
Fill in the missing chemical symbol or element name for each of the following pairs.
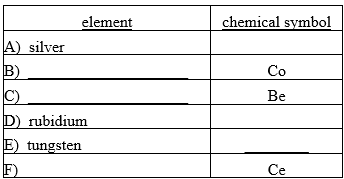
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate whether the following descriptions of matter represents an -One substance present, one phase present, substancecannot be decomposed by chemical means. ____________________
A) element
B) mixture
C) compound
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate whether each of the following statements represents a chemical change, a physical change, or no change occurred. -A red piece of copper wire is added to a beakercontaining hydrochloric acid (a clear, colorless solution).After an hour a red wire is observed in a clear,colorless solution. ____________________
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which pair does the symbol not match the name of the element?
A) nitrogen - N
B) iron - Fe
C) strontium - Sr
D) arsenic - Ar
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A pure substance has been isolated in the laboratory. Based on the two characteristics below, how can the substance be classified? I. The species cannot be separated by physical means. II. The species can be separated by chemical means.
A) an element
B) a compound
C) a homogeneous mixture
D) a heterogeneous mixture
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For each characterization of matter, choose the appropriate classification from the response list. Responses may be used more than once or need not be used at all. - 1 substance present, 1 phase present, substance can be decomposed by chemical means
A) heterogeneous mixture
B) homogeneous mixture
C) compound
D) element
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For each characterization of matter, choose the appropriate classification from the response list. Responses may be used more than once or need not be used at all. - 2 substances present, 3 phases present
A) heterogeneous mixture
B) homogeneous mixture
C) compound
D) element
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Gallium has a melting point of 30 °C and a boiling point of 2403 °C. At which temperature below will gallium be a gas?
A) -25 °C
B) 25 °C
C) 250 °C
D) 2500 °C
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a physical change?
A) burning coal
B) synthesis of methane from carbon and hydrogen
C) smoking a cigarette
D) filtering muddy water
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following sequences of elements do each of the elements have a symbol which starts with a letter not the first letter of the element's English name?
A) silver, gold, mercury
B) copper, helium, neon
C) cobalt, chromium, sodium
D) argon, iron, lead
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is an example of a homogeneous mixture?
A) chocolate chip cookie
B) a rock collection
C) glucose solution
D) oil & vinegar salad dressing
F) B) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following statements about compounds and mixtures is correct?
A) Since two or more simpler substances can be obtained from a compound, all compounds are mixtures.
B) Compounds cannot be broken down into simpler substances by chemical means.
C) Mixtures retain the properties of their individual components.
D) Compounds always have properties that are identical to those of the elements used to produce them.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the incorrect response concerning the substances listed below. I. iron II. neon III. iodized table salt IV. cola V. steam
A) III can be decomposed by chemical means.
B) III and IV represent mixtures.
C) I and II represent elements.
D) I, II, III, IV and V represent pure substances.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As two liquid solutions are added together, a yellow solid forms. This change is most likely:
A) a physical change
B) a chemical change
C) neither
D) both
F) None of the above
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which statement is true about elements?
A) Elements have similar symbols and names.
B) Element names are derived either from geographical names or from the plants.
C) The first letter of any chemical symbol is capitalized.
D) The elements whose symbols are derived from non-English names are the recently found elements.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a chemical property?
A) condensation
B) mass
C) solubility
D) burning
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate whether each of the following statements represents a chemical change, a physical change, or no change occurred. -Candle wax is burned. ____________________
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a property of both solids and liquids?
A) definite shape
B) indefinite shape
C) definite volume
D) indefinite volume
F) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Showing 1 - 20 of 75
Related Exams