B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cheeseburger from a fast food restaurant contains 19 g of fat,20 g of carbohydrate,and 28 g of protein.How many kcal of energy does the cheeseburger contain? (The caloric values are: 4 kcal/g for carbohydrate,9 kcal/g for fat,and 4 kcal/g for protein.) Give the answer to 2 significant figures.
A) 70.kcal
B) 360 kcal
C) 17 kcal
D) 630 kcal
E) 280 kcal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
One cup of kidney beans contains 15 g of protein,1 g of fat,and 42 g of carbohydrate.How many kilocalories,to two significant figures,does this sample contain? (The caloric values are: 4 kcal/g for carbohydrate,9 kcal/g for fat,and 4 kcal/g for protein.)
A) 60.kcal
B) 88 kcal
C) 230 kcal
D) 240 kcal
E) 520 kcal
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A patient has a temperature of 38.5 °C.What is the temperature in degrees Fahrenheit?
A) 70.5 °F
B) 311 °F
C) 126.9 °F
D) 101.3 °F
E) 11.7 °F
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A liquid has its own volume and shape.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A heating curve illustrates ________.
A) what a substance looks like as it is heated
B) what happens to the particles of a substance as it is heated
C) what happens to the heat applied as the temperature is increased
D) the changes in the temperature and physical state of a substance as it is heated
E) the chemical changes that occur as the substance is heated
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
A bungee jumper just before she leaps,has ________ energy.
Correct Answer

verified
Correct Answer
verified
True/False
Steam at 100 °C contains the same amount of heat as liquid water at 100 °C.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat of fusion for water is 80.cal/g.How many calories of heat are released when 20.0 g of water at 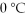 is frozen to ice?
is frozen to ice?
A) 620 cal
B) 1600 cal
C) 2000 cal
D) 2200 cal
E) 0 cal
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties describes a liquid?
A) has its own shape
B) particles are close together and move randomly
C) particles move very rapidly
D) fills the entire volume of the container
E) There is essentially no interaction between the particles.
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
Ice melting is a chemical change.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat of copper is 0.0920 cal/g °C,and the specific heat of silver is 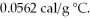 If 100 cal of heat is added to one g of each metal at 25 °C,what is the expected result?
If 100 cal of heat is added to one g of each metal at 25 °C,what is the expected result?
A) The copper will reach a higher temperature.
B) The silver will reach a higher temperature.
C) The two samples will reach the same temperature.
D) The copper will reach a temperature lower than 25 °C.
E) The silver will soften.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy of motion is referred to as ________.
A) work
B) freezing
C) specific heat
D) potential energy
E) kinetic energy
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Water vapor is a gas.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following does not represent a step on the heating curve of water?
A) The temperature of steam cannot exceed 100 °C.
B) The temperature of ice remains at 0 °C as it melts.
C) The temperature of liquid water increases linearly as it is heated.
D) The temperature of liquid water remains at 100 °C as it boils.
E) Both liquid water and ice are present at 0 °C.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not an element?
A) tin
B) water
C) gold
D) silver
E) carbon
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The temperature at which water melts and freezes is the same.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Air is a heterogeneous mixture.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A potato contains 20 g of carbohydrate.If carbohydrate has a caloric value of 4 kcal/g,how many kcal are obtained from the carbohydrate in the potato?
A) 5 kcal
B) 20 kcal
C) 40 kcal
D) 60 kcal
E) 80 kcal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Air is an example of a ________ mixture.
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 108
Related Exams