A) (s)
B) (g)
C) (l)
D) (w)
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Before a chemical equation can be written,one must know:
A) the atomic mass of all the elements involved.
B) the molar mass of all the compounds.
C) the symbols and formulas of all reactants and products.
D) the number of moles of all reactants and products.
E) none of the above.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The following equation IS balanced: 2 C4H10O + 13 O2 → 8 CO2 + 10 H2O
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methane gas (CH4) ,on complete combustion in air,produces: 1.CO2 2.H2 3.H2O
A) 1 and 2 only
B) 1 and 3 only
C) 2 and 3 only
D) All of 1 and 2 only
E) None of
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be the formula of the precipitate that forms when Pb(NO3) 2 (aq) and K2SO4 (aq) are mixed?
A) K(NO3) 2
B) PbSO4
C) PbK2
D) H2O
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you had an aqueous mixture that contained Ag+,K+,and Pb2+ cations,how many different solids could precipitate if a chloride solution was added?
A) 1
B) 2
C) 3
D) 4
E) no solids will precipitate
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about balancing reactions is FALSE?
A) If there is no coefficient or subscript,a one is implied.
B) When coefficients are added the type of compounds is changed in the chemical reaction.
C) Subscripts are multiplied by the coefficients to determine the number of atoms in a compound.
D) Coefficients are added to compounds to ensure both sides of the reaction have equal numbers of each atom.
E) All of the above statements are true.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is NOT evidence that a chemical reaction has occurred?
A) color change when chemicals are contacted with each other
B) solid formation when chemicals are contacted with each other
C) gas formation when chemicals are contacted with each other
D) emission of light when chemicals are contact with each other
E) All of the above are evidence of a chemical reaction.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The following reaction IS balanced: AgNO3 + 2NH4Cl → AgCl + (NH4)2NO3
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of a reaction occurs when a hydrochloric acid solution is mixed with a sodium bicarbonate solution?
A) precipitation
B) acid-base neutralization
C) oxidation-reduction
D) gas evolution
E) no reaction
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The net ionic equation for the reaction of sodium hydroxide plus hydrochloric acid is 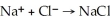 .
.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An aqueous solution is:
A) any liquid with another compound dissolved in it.
B) an ionic compound with water dissolved in it.
C) water with a molecular compound dissolved in it.
D) water with another compound dissolved in it.
E) none of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
One of the characteristics of an acid-base reaction is that this type of reaction forms water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following equations is NOT balanced properly?
A) 2Cr + 6HCl → 2CrCl3 + 3H2
B) 2NaHCO3 → Na2CO3 + CO2 + H2O
C) Cr2(SO4) 3 + 6KOH → 2Cr(OH) 3 + 3 K2SO4
D) 4NH3 + 14O2 → 4NO2 + 6H2O
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Considering the following precipitation reaction: Pb(NO3) 2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq) What is the correct net ionic equation?
A) Pb2+ + I2- → PbI2(s)
B) 2NO3- + 2K+ → 2KNO3
C) Pb2+ + 2NO3- + 2K+ + 2I- → PbI2(s) + 2K+ + 2NO3-
D) Pb2+ + 2I- → PbI2(s)
E) none of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All of the following compounds are soluble in water EXCEPT:
A) NaCl.
B) CaCl2.
C) FeCl3.
D) NH4Cl.
E) PbCl2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a solution of MgCl2 and one of AgNO3 are mixed,the net ionic equation is:
A) Mg2+(aq) + 2Cl-(aq) + 2Ag+(aq) + 2NO3- (aq) → Mg2+(aq) + 2NO3-aq) + 2AgCl(s)
B) Mg2+(aq) + 2Cl-(aq) + 2Ag+(aq) + 2NO3- (aq) → Mg(NO3) 2 (aq) + 2 AgCl(s)
C) Mg2+(aq) + 2NO3-(aq) → Mg(NO3) 2(aq)
D) Cl-(aq) + Ag+(aq) → AgCl(s)
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of a reaction occurs when a sodium hydroxide solution is mixed with an acetic acid solution?
A) precipitation
B) acid-base neutralization
C) oxidation-reduction
D) gas evolution
E) no reaction
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the coefficients for the following reaction when it is properly balanced? ___potassium iodide + ___lead (II) acetate → ___lead (II) iodide +___potassium acetate
A) 2,1,1,1
B) 2,1,1,2
C) 3,2,2,1
D) 1,1,2,2
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A reaction which forms a solid product is an example of a(n) :
A) oxidation-reduction reaction.
B) combustion reaction.
C) precipitation reaction.
D) gas evolution reaction.
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 113
Related Exams