A) 34.0 mol
B) 27.2 mol
C) 6.80 mol
D) 5.44 mol
E) 2.27 mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrochloric acid can be prepared by the following reaction: 2NaCl(s) + H2SO4(aq) → 2HCl(g) + Na2SO4(s) What mass of HCl can be prepared from 2.00 mol H2SO4 and 150.g NaCl?
A) 2.57 g
B) 46.8 g
C) 93.6g
D) 146 g
E) 167g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the theoretical yield of vanadium that can be produced by the reaction of 40.0 g of V2O5 with 40.0 g of calcium based on the following chemical equation? V2O5(s) + 5Ca(l) → 2V(l) + 5CaO(s)
A) 11.2 g
B) 5.6 g
C) 22.4 g
D) 40.0 g
E) 20.3 g
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of nitrogen gas is required to react completely with excess hydrogen gas to produce 13.6 g of ammonia?
A) 11.2 g
B) 0.06 g
C) 22.4 g
D) 16.5 g
E) 44.8 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given that CaO(s) + H2O(l) → Ca(OH) 2(s) , heat of reaction = −64.8 kJ/mol, how many grams of CaO must react in order to liberate 525 kJ of heat?
A) 6.92 g
B) 56.1 g
C) 454 g
D) 606 g
E) 3.40× 104 g
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many grams of Cl2 can be prepared from the reaction of 16.0 g of MnO2 and 30.0 g of HCl according to the following balanced chemical equation? MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O
A) 6.52 g
B) 7.29 g
C) 13.0 g
D) 14.6 g
E) 58.4 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The highly exothermic thermite reaction, in which aluminum reduces iron(III) oxide to elemental iron, has been used by railroad repair crews to weld rails together. 2Al(s) + Fe2O3(s) → 2Fe(s) + Al2O3(s) heat of reaction = −847.6 kJ/mol What mass of iron is formed if 725 kJ of heat are released?
A) 23.9 g
B) 47.8 g
C) 65.3 g
D) 95.5 g
E) 112 g
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum reacts with oxygen to produce aluminum oxide which can be used as an adsorbent, desiccant or catalyst for organic reactions.A mixture of 82.49 g of aluminum and 117.65 g of oxygen is allowed to react.Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.
A) Oxygen is the limiting reactant; 19.81 g of aluminum remain.
B) Oxygen is the limiting reactant; 35.16 g of aluminum remain.
C) Aluminum is the limiting reactant; 16.70 g of oxygen remain.
D) Aluminum is the limiting reactant; 35.16 g of oxygen remain.
E) Aluminum is the limiting reactant; 44.27 g of oxygen remain.
G) C) and D)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
What is the volume of NH3 produced in the following reaction when 3.0 L of N2 reacts with 4.0 L of H2 ? N2(g) + 3H2(g) → 2NH3(g)
A) 1.5 L
B) 2.7 L
C) 6.0 L
D) 7.5 L
E) 12 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of 0.200 M H2SO4 solution is required to react exactly with 50.0 mL of 0.100 M KOH? H2SO4(aq) + 2KOH(aq) → 2H2O(l) + K2SO4(aq)
A) 62.5 mL
B) 12.5 mL
C) 50.0 mL
D) 25.0 mL
E) 75.0 mL
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 3.41 g of nitrogen react with 2.79 g of hydrogen to produce ammonia, what is the limiting reactant and what mass of ammonia is produced?
A) Hydrogen is the limiting reactant and 0.22 g of ammonia are produced.
B) Nitrogen is the limiting reactant and 2.07 g of ammonia are produced.
C) Nitrogen is the limiting reactant and 4.15 g of ammonia are produced.
D) Hydrogen is the limiting reactant and 23.5 g of ammonia are produced.
E) Hydrogen is the limiting reactant and 15.8 g of ammonia are produced.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum metal reacts with chlorine gas to form solid aluminum trichloride.What mass of chlorine gas is required to react completely with 163 g of aluminum?
A) 214 g
B) 286g
C) 321 g
D) 428 g
E) 643 g
G) B) and D)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Consider the following reaction.H2(g) + I2(s) → 2 HI(g) If the cylinder with a movable piston below contains 1 mol H2(g) and 1 mol I2(s) , which represents the cylinder after the reaction is complete? 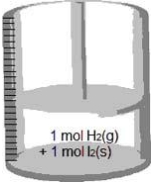
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When active metals such as magnesium are immersed in acid solution, hydrogen gas is evolved.Calculate the volume of H2(g) at 30.1°C and 0.85 atm that can be formed when 275 mL of 0.725 M HCl solution reacts with excess Mg to give hydrogen gas and aqueous magnesium chloride. Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) (R = 0.08206 L • atm/K • mol)
A) 3.4 × 10−3 L
B) 2.2 L
C) 2.9 L
D) 5.8 L
E) 11.7 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of grams of ammonia, NH3, which can be obtained from the reaction of 10.0 g of H2 and 80.0 g of N2? N2 + 3H2 → 2NH3
A) 34.1 g
B) 48.6 g
C) 56.3 g
D) 90.0 g
E) 97.3 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the theoretical yield of chromium that can be produced by the reaction of 40.0 g of Cr2O3 with 8.00 g of aluminum according to the chemical equation below? 2Al + Cr2O3 → Al2O3 + 2Cr
A) 7.7 g
B) 15.4 g
C) 27.4 g
D) 30.8 g
E) 49.9 g
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tetraphosphorus hexaoxide is formed by the reaction of phosphorus with oxygen gas.If a mixture of 75.3 g of phosphorus and 38.7 g of oxygen produce 43.3 g of P4O6, what is the percent yield for the reaction?
A) 57.5%
B) 48.8%
C) 38.0%
D) 89.4%
E) 16.3%
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methanol (CH3OH) is converted to bromomethane (CH3Br) as follows: CH3OH + HBr → CH3Br + H2O If 12.23 g of bromomethane are produced when 5.00 g of methanol is reacted with excess HBr, what is the percentage yield?
A) 12.9%
B) 33.8%
C) 40.9%
D) 59.1%
E) 82.6%
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the theoretical yield of vanadium, in moles, that can be produced by the reaction of 2.0 mole of V2O5 with 6.0 mole of calcium based on the following chemical equation? V2O5(s) + 5Ca(l) → 2V(l) + 5CaO(s)
A) 1.0 mol
B) 1.6 mol
C) 2.0 mol
D) 2.4 mol
E) 4.0 mol
G) C) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
How many milliliters of 0.0150 M KOH are required to react exactly with 75.0 mL of 0.0150 M H3PO4? H3PO4(aq) + 3KOH(aq) → K3PO4(aq) + 3H2O(l)
A) 25.0 mL
B) 1000 mL
C) 225 mL
D) 250 mL
E) 75.0 mL
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 61
Related Exams