Correct Answer

verified
Correct Answer
verified
Short Answer
Of the given pair of compounds, which would have the higher boiling point? C3H8 or CH3OCH3
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Silver metal crystallizes in a face-centered cubic lattice with L as the length of one edge of the unit cube. The center-to-center distance between nearest silver atoms is
A) L/2.
B) ![]()
C) 2L.
D) ![]()
E) None of the above.
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
The molar enthalpy of vaporization of carbon disulfide is 26.74 kJ/mol, and its normal boiling point is 46°C. What is the vapor pressure of CS2 at 0°C?
A) 447 torr
B) 4160 torr
C) 313 torr
D) 139 torr
E) 5.47 torr
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Crystals of elemental sulfur are easily crushed, and melt at 113°C. Liquid sulfur does not conduct electricity. What kind of crystal is this?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The vapor pressure of ethanol is 400 mmHg at 63.5°C. Its molar heat of vaporization is 39.3 kJ/mol. What is vapor pressure of ethanol, in mmHg, at 34.9°C?
A) 1,510 mmHg
B) 100 mmHg
C) 200 mmHg
D) 0.0099 mmHg
E) 4.61 mmHg
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances should have the highest boiling point?
A) CH4
B) Cl2
C) Kr
D) CH3Cl
E) N2
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Short Answer
Suppose the atoms in a two-dimensional crystal have the following arrangement: 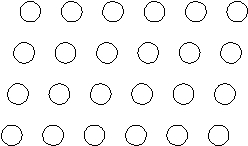 What is the coordination number of each atom in this crystal?
What is the coordination number of each atom in this crystal?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be expected to have the highest vapor pressure at room temperature?
A) ethanol, bp = 78°C
B) methanol, bp = 65°C
C) water, bp = 100°C
D) acetone, bp = 56°C
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Methane has a heat of fusion of 0.84 kJ/mol and a heat of vaporization of 9.2 kJ/mol. Estimate the value for the heat of sublimation.
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the pair of compounds given, which would have the stronger intermolecular forces of attraction? NH3 or PH3
Correct Answer

verified
Correct Answer
verified
Short Answer
Iron crystallizes in a body-centered cubic unit. The edge of this cell is 287 pm. Calculate the density of iron.
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in CH3OH(l).
Correct Answer

verified
hydrogen bonding and dispersion
Correct Answer
verified
Short Answer
Of the given pair of compounds, which would have the higher boiling point? NH3 or CH4
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following compound and its boiling point, identify whether it is polar or nonpolar: F2, -188.1°C.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Krypton has a higher melting point than argon because of its
A) hydrogen bonding.
B) stronger dispersion forces.
C) permanent dipole moment.
D) ionic bonds.
E) greater ionization energy.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A liquid boils when its
A) vapor pressure is exactly 1 atmosphere.
B) vapor pressure is equal to, or greater than, the external pressure pushing on it.
C) temperature is equal to 273 K (standard temperature) .
D) temperature is greater than room temperature.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms does not participate in hydrogen bonding?
A) S
B) O
C) F
D) N
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
MgO has the same crystal structure as NaCl, face-centered cubic. How many oxide ions surround each Mg2+ ion as nearest neighbors?
A) 4
B) 6
C) 8
D) 10
E) 12
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The normal boiling point of bromine is 58.8°C. Given that the vapor pressure of bromine is 75.0 torr at 2.5°C, calculate the molar enthalpy of vaporization of bromine.
A) 3.76 kJ/mol
B) 31.3 kJ/mol
C) 2.90 kJ/mol
D) 3.57 kJ/mol
E) 29.7 kJ/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 138
Related Exams