A) [He] 2s2 + e- → [He] 2s2 2p1
B) [He] 2s2 2p2 + e- → [He] 2s2 2p3
C) [He] 2s2 2p3 + e- → [He] 2s2 2p4
D) [He] 2s2 2p6 + e- → [He] 2s2 2p6 3s1
F) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Using shorthand notation,the ground-state electron configuration of the platinum ion in Cs2Pt is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Metals tend to react with the halogens to form metal halides.What is the reactivity order for the halogens?
A) F2 > Cl2 > Br2 > I2
B) Cl2 > F2 > Br2 > I2
C) Br2 > I2 > Cl2 > F2
D) I2 > Br2 > Cl2 > F2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are in the outermost shell of the In3+ ion in its ground state?
A) 2
B) 3
C) 6
D) 18
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four spheres below represent Na+,Mg2+,F⁻,and O2-,not necessarily in that order. 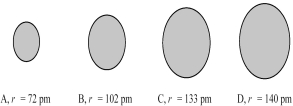 -Which sphere most likely represents the Na⁺ ion?
-Which sphere most likely represents the Na⁺ ion?
A) A
B) B
C) C
D) D
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate which is larger in each of the following two sets. (I) Cr3+ or Cr (II) Se2- or Se
A) Cr3+ is larger than Cr and Se2- is larger than Se.
B) Cr3+ is larger than Cr and Se is larger than Se2-.
C) Cr is larger than Cr3+ and Se2- is larger than Se.
D) Cr is larger than Cr3+ and Se is larger than Se2-.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
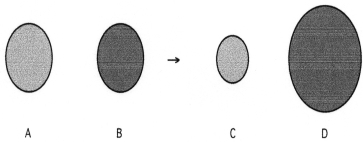 -Which sphere represents the monatomic anion?
-Which sphere represents the monatomic anion?
A) A
B) B
C) C
D) D
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Isoelectronic means having the same number of electrons.The dipositive ion that is isoelectronic with Br- is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following pictures represent alkali halide salts. 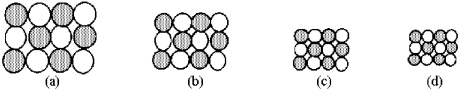 -Which salt has the highest lattice energy?
-Which salt has the highest lattice energy?
A) picture (a)
B) picture (b)
C) picture (c)
D) picture (d)
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ionic compound would be expected to have the highest lattice energy?
A) Rb2O
B) Sr O
C) In2O3
D) C O2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The number of electrons in the ion P3- is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms of which element,indicated by letter on the periodic table,would be expected to have the most negative value of Eea? 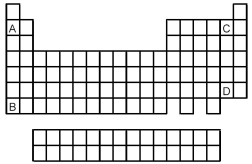
A) A
B) B
C) C
D) D
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
If niobium loses all of its valence electrons when it reacts with fluorine,what is the formula of the neutral binary compound that results?
Correct Answer

verified
Correct Answer
verified
Short Answer
When the equation for the reaction of KBr(aq)with MnO2(s)to produce Br2 and Mn2+(aq)in acidic solution is balanced,the coefficient in front of the Br2 is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
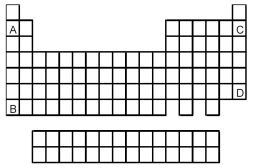 -Atoms of which element,indicated by letter on the periodic table,is expected to have the smallest atomic radius?
-Atoms of which element,indicated by letter on the periodic table,is expected to have the smallest atomic radius?
A) A
B) B
C) C
D) D
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not a chemical reaction of the alkali metal potassium?
A) K(s) + O2(g) → KO2(s)
B) 2 K(s) + 2 H2O(l) → 2 KOH(aq) + H2(g)
C) 2 K(s) + H2(g) → 2 KH(s)
D) 6 K(s) + N2(g) → 2 K3N(s)
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Group 2A metals tend to be somewhat less reactive than alkali metals,and the order of their reactivity is
A) Ba > Sr > Ca > Mg > Be.
B) Be > Mg > Ca > Sr > Ba.
C) Ca > Mg > Be > Ba > Sr.
D) Sr > Ca > Mg > Be > Ba.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An incorrect statement about the alkaline earth metals is:
A) Melting points generally decrease as one descends the group.
B) Densities are less than those of the corresponding alkali elements of the same period.
C) Ionic radii of the M2+ ion increases as one descends the group.
D) The first ionization energy is less than that of the second ionization energy.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ion does not have a noble gas configuration in its ground state?
A) Sc3+
B) Al3+
C) Ga3+
D) As3-
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons does magnesium lose and nitrogen need to form Mg3N2?
A) magnesium loses 2 and nitrogen gains 2
B) magnesium loses 2 and nitrogen gains 3
C) magnesium loses 3 and nitrogen gains 2
D) magnesium loses 3 and nitrogen gains 3
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 173
Related Exams