Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in CCl4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ibuprofen is used as an analgesic for the relief of pain, and also to help reduce fever.What is the hybridization state of carbon indicated by the arrow in the structure of ibuprofen shown below? 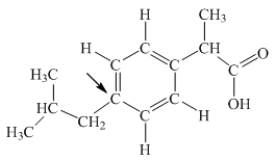
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A central atom with 5 electron pairs (single bonds and/or lone pairs of electrons) could have which of the following molecular geometries? I.Trigonal bipyramidal II.Seesaw III.T-shaped IV.Linear
A) I, II, and III
B) II, III, and IV
C) I, III, and IV
D) I, II, and IV
E) I, II, III, and IV
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An sp2 hybridized central carbon atom with no lone pairs of electrons has what type of bonding?
A) 1 and 2 bonds
B) 1 and 3 bonds
C) 2 and 2 bonds
D) 3 and 2 bonds
E) 0 and 4 bonds
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
N,N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents.What is the hybridization state of carbon indicated by the arrow in the structure of DEET shown below? 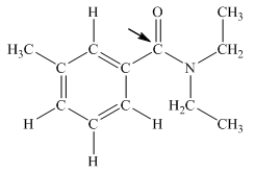
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp2 hybridized terminal oxygen atom (bonded to one other atom only) with 2 lone pairs of electrons has what type of bonding?
A) 1 and 1 bonds
B) 2 and 1 bonds
C) 1 and 2 bonds
D) 0 and 2 bonds
E) 0 and 3 bonds
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the molecular geometry of IF5.
A) 0 lone pairs, square pyramidal
B) 0 lone pairs, trigonal bipyramidal
C) 1 lone pair, octahedral
D) 1 lone pair, square pyramidal
E) 2 lone pairs, pentagonal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to Valence Bond Theory which orbital is left vacant in the molecule BF3?
A) sp3
B) sp2
C) sp
D) p
E) s
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond angle in Cl2O is expected to be approximately
A) 90°
B) 109.5°
C) 120°
D) 145°
E) 180°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate the number of -bonds in C2H4.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp3d hybridized central atom has what angles between its hybrid orbitals?
A) 120° only
B) 120° and 109.5°
C) 109.5° only
D) 120° and 90°
E) < 90°only
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A bonding molecular orbital is of lower energy (more stable)than the atomic orbitals from which it was formed.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the molecular geometry and polarity of the SO2 molecule.
A) linear, polar
B) linear, nonpolar
C) bent, polar
D) bent, nonpolar
E) None of the above.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following would the bonding be weakened with the subtraction of an electron to form a positive molecular ion?
A) N2
B) O2
C) F2
D) H2
E) Cl2
G) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Consider the species N2-, N2, and N2+.Which of these species will be paramagnetic?
A) N2 and N2-
B) N2+ and N2
C) N2+ and N2-
D) only N2-
E) none are paramagnetic
G) C) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
What is the bond order of Cl2+
A) 0
B) 0.5
C) 1
D) 1.5
E) 2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sp2 hybridized central atom has what angles between its hybrid orbitals?
A) 145°
B) 120°
C) 109.5°
D) 90°
E) < 90°
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the species Cl2+, Cl2, and Cl2-.Which of these species will be paramagnetic?
A) only Cl2
B) Cl2+ and Cl2
C) Cl2 and Cl2-
D) Cl2+ and Cl2-
E) all three are paramagnetic
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of pi bonds in the oxalate ion (C2O42-) is
A) 1
B) 2
C) 3
D) 4
E) 5
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 147
Related Exams