B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about fission and fusion is false?
A) Fission occurs among the heaviest isotopes, whereas fusion occurs more readily for light isotopes.
B) For a fission reaction the mass defect ( m) is negative, whereas for fusion m is positive.
C) In order for fusion reactions to occur, temperatures must be in the millions of degrees.
D) The fission of Pu-239 atoms produces a great number of isotopes of a large number of elements.
E) Neutron-induced fission processes can occur at room temperature, rather than at millions of degrees.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Radium-226 decays by alpha emission.What is its decay product?
A) ![]() Fr
Fr
B) ![]() Rn
Rn
C) ![]() Th
Th
D) ![]() Ac
Ac
E) ![]() Po
Po
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Balance the equation 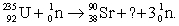
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which isotope, when bombarded with oxygen-18, yields the artificial isotope seaborgium-263 plus 4 neutrons?
A) nobelium-245
B) radium-259
C) californium-245
D) nobelium-249
E) californium-249
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The fundamental unit of radioactivity is the ______ (Ci)
Correct Answer

verified
Correct Answer
verified
Short Answer
Tritium is a radioisotope of hydrogen having a half-life of 12.3 years.If you initially had 1.0 × 10-7 mole of tritium, calculate the decay rate of the sample.
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict which nucleus is less stable, 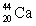 or
or 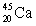 .
.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the energy released in joules when one mole of polonium-214 decays according to the equation ![Calculate the energy released in joules when one mole of polonium-214 decays according to the equation Po Pb + He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.] A) 8.78 × 10<sup>14</sup> J/mol B) 7.2 × 10<sup>14</sup> J/mol C) 8.78 × 10<sup>11</sup> J/mol D) -9.75 × 10<sup>-3</sup> J/mol E) 1.46 × 10<sup>-9</sup> J/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f3d_efac_a2ab_659e81ceb826_TB3246_11.jpg) Po
Po ![Calculate the energy released in joules when one mole of polonium-214 decays according to the equation Po Pb + He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.] A) 8.78 × 10<sup>14</sup> J/mol B) 7.2 × 10<sup>14</sup> J/mol C) 8.78 × 10<sup>11</sup> J/mol D) -9.75 × 10<sup>-3</sup> J/mol E) 1.46 × 10<sup>-9</sup> J/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f3d_efad_a2ab_51c2383d410c_TB3246_11.jpg)
![Calculate the energy released in joules when one mole of polonium-214 decays according to the equation Po Pb + He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.] A) 8.78 × 10<sup>14</sup> J/mol B) 7.2 × 10<sup>14</sup> J/mol C) 8.78 × 10<sup>11</sup> J/mol D) -9.75 × 10<sup>-3</sup> J/mol E) 1.46 × 10<sup>-9</sup> J/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f3e_16be_a2ab_99924e5bfde6_TB3246_11.jpg) Pb +
Pb + ![Calculate the energy released in joules when one mole of polonium-214 decays according to the equation Po Pb + He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.] A) 8.78 × 10<sup>14</sup> J/mol B) 7.2 × 10<sup>14</sup> J/mol C) 8.78 × 10<sup>11</sup> J/mol D) -9.75 × 10<sup>-3</sup> J/mol E) 1.46 × 10<sup>-9</sup> J/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3246/11ea7cbf_8f3e_16bf_a2ab_edc130e4adfd_TB3246_11.jpg) He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.]
He. [Atomic masses: Pb-210 = 209.98284 amu, Po-214 = 213.99519 amu, He-4 = 4.00260 amu.]
A) 8.78 × 1014 J/mol
B) 7.2 × 1014 J/mol
C) 8.78 × 1011 J/mol
D) -9.75 × 10-3 J/mol
E) 1.46 × 10-9 J/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sulfur-35 decays by beta emission.The decay product is
A) ![]() P
P
B) ![]() S
S
C) ![]() Si
Si
D) ![]() Cl
Cl
E) ![]() Cl
Cl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 24% of a certain radioisotope decays in 6.5 years, what is the half-life of this isotope?
A) 3.9 yr
B) 16 yr
C) 0.22 yr
D) 2.2 yr
E) 3.2 yr
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Present-day plant life has a carbon-14 decay rate of 16 disintegrations per minute (dpm) per gram of carbon.If a contemporary wooden chair were somehow preserved for the next 3,900 years, what 14C decay rate should be expected from the wood used to make the chair? (t1/2 = 5,730 yr)
A) 26 dpm
B) 12 dpm
C) 11 dpm
D) 10 dpm
E) 8 dpm
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Estimate the age of a bottled wine that has a tritium, 3H, content 60% that of freshly bottled wine.Tritium decays by beta decay and has a half-life of 12.3 yr. 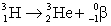
A) 0.029 yr
B) 7.4 yr
C) 9.1 yr
D) 16 yr
E) 35 yr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When atoms of aluminum-27 are bombarded with alpha particles, a neutron and an element are produced.The particular isotope formed is 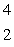 He +
He + 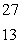 Al
Al 
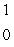 n + ____
n + ____
A) ![]() Si
Si
B) ![]() Si
Si
C) ![]() S
S
D) ![]() P
P
E) ![]() P
P
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What type of nuclear process is illustrated below? 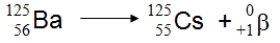
Correct Answer

verified
Correct Answer
verified
True/False
Naturally occurring uranium contains approximately 1% U-235 and 99% U-238.Of these, the isotope that undergoes fission in a nuclear reactor is U-238.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alpha particles are identical to
A) protons.
B) helium atoms.
C) hydrogen atoms.
D) helium nuclei.
E) electrons.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The only stable isotope of iodine is iodine-127.Predict the mode of decay of 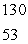 I.
I.
A) alpha emission
B) beta emission
C) positron emission
D) electron capture
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List the given types of nuclear radiation (cosmic rays, medical and dental X rays, and nuclear waste) in order of how much each contributes to the average yearly dose of nuclear radiation for AmericAns :
A) cosmic rays < medical and dental X rays < nuclear waste
B) medical and dental X rays < cosmic rays < nuclear waste
C) nuclear waste < cosmic rays < medical and dental X rays
D) cosmic rays < nuclear waste < medical and dental X rays
E) medical and dental X rays < nuclear waste < cosmic rays
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rubidium-87 decays by beta decay with a half-life of 4.9 × 1010 yr.How many 87Rb atoms are in a moon rock sample that has a rubidium decay rate of 3,500 disintegrations per hour?
A) 9.0 × 1016 atoms
B) 4.3 × 10-4 atoms
C) 2.2 × 1018 atoms
D) 2.5 × 1014 atoms
E) 1.7 × 1014 atoms
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 133
Related Exams