A) I
B) II
C) III
D) IV
E) No reaction will occur.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following functional groups can be hydrolyzed to a carbonyl using excess water in acid?
A) Imine
B) Enamine
C) Acetal
D) Hemiacetal
E) All of the above (a-d)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the aldol condensation product for the following reaction? 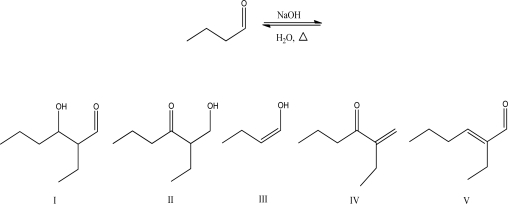
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the aldol condensation product for the following reaction? 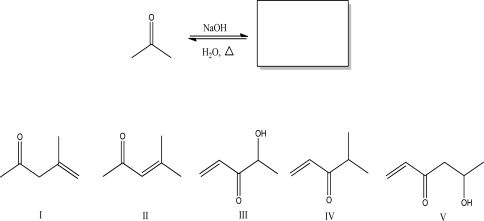
A) I
B) II
C) III
D) IV
E) V
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Provide the starting material for the following reaction. 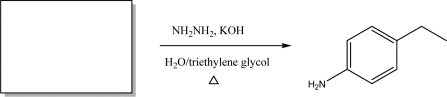
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following nucleophiles would add to an α,β-unsaturated ketone via direct addition?
A) KCN
B) KOH
C) Br2
D) NH2CH2CH2CH3
E) HOCH2CH2CH3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the product and provide a mechanism for the following reaction. 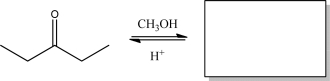
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the product for the following reaction. 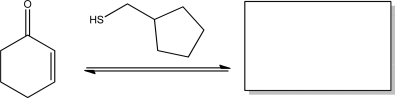
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the most likely product of the following reaction? 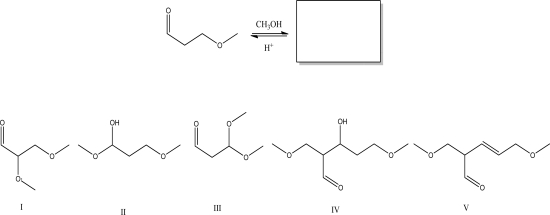
A) I
B) II
C) III
D) IV
E) V
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product for the following reaction? 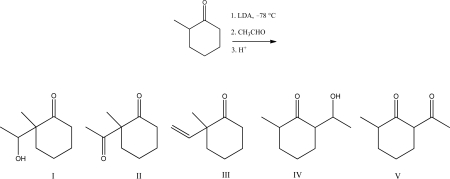
A) I
B) II
C) III
D) IV
E) V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules cannot undergo an intramolecular aldol condensation when treated with NaOH? 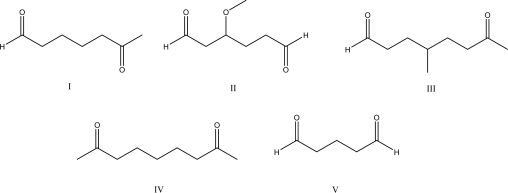
A) I
B) II
C) III
D) IV
E) V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the carbonyl of an aldehyde is protonated with H2SO4,which of the following is not true?
A) The protonated aldehyde is more likely to react with alcohols.
B) The protonated carbonyl has a formal charge.
C) The protonated aldehyde becomes more nucleophilic.
D) The protonation of the aldehyde is reversible.
E) The protonated aldehyde is more likely to react with water.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the appropriate starting material for the reaction below. 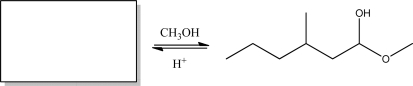
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the aldol condensation product for the following reaction? 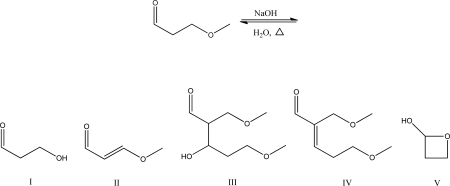
A) I
B) II
C) III
D) IV
E) V
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true of the Wolff-Kishner reduction?
A) It always takes place under acidic conditions.
B) It converts ketones to alcohols.
C) It converts aldehydes to alcohols.
D) It converts carbonyls in ketones to a CH2 group.
E) It requires neutral conditions.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following functional groups is present in the compound below? 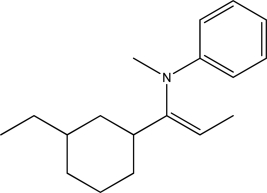
A) Amine
B) Imine
C) Enamine
D) Amide
E) Acetal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the most likely product of the following reaction? 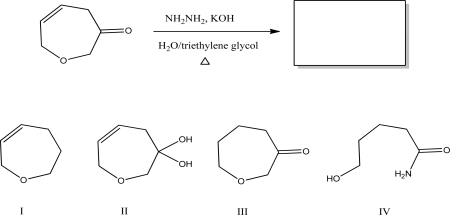
A) I
B) II
C) III
D) IV
E) No reaction will occur.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the starting material that best completes the following reaction. 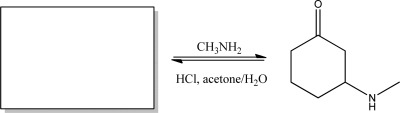
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the most likely product of the following reaction? 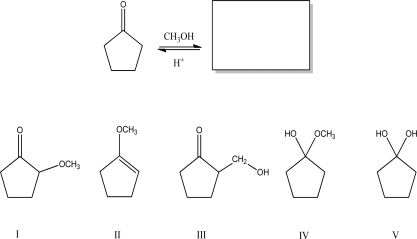
A) I
B) II
C) III
D) IV
E) V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the most likely product of the following reaction? 
A) I
B) II
C) III
D) IV
E) V
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 63
Related Exams