A) 10.0 g hydrogen remains
B) 10.9 g hydrogen remains
C) 15.0 g hydrogen remains
D) 19.9 g hydrogen remains
E) Need to know the equilibrium constant in order to calculate the answer.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is at equilibrium at a pressure of 1 atm, in a closed container. NaOH(s) + CO2(g)  NaHCO3(s) H°rxn < 0
Which, if any, of the following actions will decrease the concentration of CO2 gas present at equilibrium?
NaHCO3(s) H°rxn < 0
Which, if any, of the following actions will decrease the concentration of CO2 gas present at equilibrium?
A) adding N2 gas to double the pressure
B) adding more solid NaOH
C) increasing the volume of the container
D) lowering the temperature
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Ammonia is synthesized in the Haber process:
N2(g) + 3H2(g)  2NH3(g)
Kp for this reaction is 1.49 × 10-5 at 500.°C. Calculate Kc at this temperature.
2NH3(g)
Kp for this reaction is 1.49 × 10-5 at 500.°C. Calculate Kc at this temperature.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the mass-action expression, Qc , for the following chemical reaction. Zn(s) + 2Ag+(aq)  Zn2+(aq) + 2Ag(s)
Zn2+(aq) + 2Ag(s)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
If Q > K, more products need to be formed as the reaction proceeds to equilibrium.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
SO2 reacts with O2 to produce SO3. If 86.0 g of SO2 is placed in a reaction vessel along with excess oxygen gas, how many moles of SO2 remain when 50.0 g of SO3 have been formed?
A) 0.56 mol
B) 0.62 mol
C) 0.72 mol
D) 0.78 mol
E) 1.34 mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the mass-action expression, Qc, for the following chemical reaction equation. 2C6H6(g) + 15O2(g)  12CO2(g) + 6H2O(g)
12CO2(g) + 6H2O(g)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a chemical reaction, if the starting concentrations of reactants are increased, then the equilibrium constant Kc will also increase.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant Kc for the reaction PCl3(g) + Cl2(g)  PCl5(g)
Is 49 at 230°C. If 0.70 mol of PCl3 is added to 0.70 mol of Cl2 in a 1.00-L reaction vessel at 230°C, what is the concentration of PCl3 when equilibrium has been established?
PCl5(g)
Is 49 at 230°C. If 0.70 mol of PCl3 is added to 0.70 mol of Cl2 in a 1.00-L reaction vessel at 230°C, what is the concentration of PCl3 when equilibrium has been established?
A) 0.049 M
B) 0.11 M
C) 0.30 M
D) 0.59 M
E) 0.83 M
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass-action expression, Qp, for the following reaction? SbF5(g) + 4Cl2(g)  SbCl3(g) + 5ClF(g)
SbCl3(g) + 5ClF(g)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of the above is the correct mass-action expression.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction, in CCl4 solvent, has been studied at 25°C. 2BrCl  Br2 + Cl2
The equilibrium constant Kc is known to be 0.141. If the initial concentration of chlorine is 0.0300 M and of bromine monochloride is 0.0200 M, what is the equilibrium concentration of bromine?
Br2 + Cl2
The equilibrium constant Kc is known to be 0.141. If the initial concentration of chlorine is 0.0300 M and of bromine monochloride is 0.0200 M, what is the equilibrium concentration of bromine?
A) 1.35 × 10-3 M
B) 2.70 × 10-3 M
C) 8.82 × 10-3 M
D) 9.70 × 10-2 M
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The equilibrium constant for reaction (1) below is 276. Under the same conditions, what is the equilibrium constant of reaction (2) ? (1) 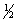 X2(g) +
X2(g) + 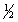 Y2(g)
Y2(g)  XY(g)
(2) 2XY(g)
XY(g)
(2) 2XY(g)  X2(g) + Y2(g)
X2(g) + Y2(g)
A) 6.02 × 10-2
B) 7.25 × 10-3
C) 3.62 × 10-3
D) 1.31 × 10-5
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 450°C, tert-butyl alcohol decomposes into water and isobutene. (CH3) 3COH(g)  (CH3) 2CCH2(g) + H2O(g)
A reaction vessel contains these compounds at equilibrium. What will happen if the volume of the container is reduced by 50% at constant temperature?
(CH3) 2CCH2(g) + H2O(g)
A reaction vessel contains these compounds at equilibrium. What will happen if the volume of the container is reduced by 50% at constant temperature?
A) The forward reaction will proceed to reestablish equilibrium.
B) The reverse reaction will proceed to reestablish equilibrium.
C) No change occurs.
D) The equilibrium constant will increase.
E) The equilibrium constant will decrease.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Stearic acid, nature's most common fatty acid, dimerizes when dissolved in hexane: 2C17H35COOH  (C17H35COOH) 2 H°rxn = -172 kJ
The equilibrium constant for this reaction at 28°C is 2900. Estimate the equilibrium constant at 38°C.
(C17H35COOH) 2 H°rxn = -172 kJ
The equilibrium constant for this reaction at 28°C is 2900. Estimate the equilibrium constant at 38°C.
A) 4.7 × 105
B) 2.6 × 104
C) 1.9 × 103
D) 3.2 × 102
E) 18
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium hydrogen carbonate decomposes above 110°C to form sodium carbonate, water, and carbon dioxide. 2NaHCO3(s)  Na2CO3(s) + H2O(g) + CO2(g)
One thousand grams of sodium hydrogen carbonate are added to a reaction vessel, the temperature is increased to 200°C, and the system comes to equilibrium. What happens in this system if another 50 g of sodium carbonate are now added?
Na2CO3(s) + H2O(g) + CO2(g)
One thousand grams of sodium hydrogen carbonate are added to a reaction vessel, the temperature is increased to 200°C, and the system comes to equilibrium. What happens in this system if another 50 g of sodium carbonate are now added?
A) The partial pressure of carbon dioxide will increase.
B) The partial pressure of carbon dioxide will decrease.
C) The partial pressure of carbon dioxide will be unchanged.
D) The amounts of all products will be greater when equilibrium is reestablished.
E) None of the above conclusions is justified without knowing the equilibrium constant.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction system CS2(g) + 4H2(g)  CH4(g) + 2H2S(g)
Is at equilibrium. Which of the following statements describes the behavior of the system if the partial pressure of hydrogen is doubled?
CH4(g) + 2H2S(g)
Is at equilibrium. Which of the following statements describes the behavior of the system if the partial pressure of hydrogen is doubled?
A) As equilibrium is reestablished, the partial pressure of carbon disulfide increases.
B) As equilibrium is reestablished, the partial pressure of methane, CH4, decreases.
C) As equilibrium is reestablished, the partial pressure of hydrogen decreases.
D) As equilibrium is reestablished, the partial pressure of hydrogen sulfide decreases.
E) As equilibrium is reestablished, all the partial pressures will decrease.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitric oxide and bromine were allowed to react in a sealed container. When equilibrium was reached PNO = 0.526 atm, 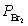 = 1.59 atm, and PNOBr = 7.68 atm. Calculate Kp for the reaction. 2NO(g) + Br2(g)
= 1.59 atm, and PNOBr = 7.68 atm. Calculate Kp for the reaction. 2NO(g) + Br2(g)  2NOBr(g)
2NOBr(g)
A) 7.45 × 10-3
B) 0.109
C) 9.18
D) 91.8
E) 134
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A good catalyst for a reaction will speed up the forward reaction and slow down the reverse reaction.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At high temperatures, carbon reacts with O2 to produce CO as follows: C(s) + O2(g)  2CO(g) .
When 0.350 mol of O2 and excess carbon were placed in a 5.00-L container and heated, the equilibrium concentration of CO was found to be 0.060 M. What is the equilibrium constant, Kc, for this reaction?
2CO(g) .
When 0.350 mol of O2 and excess carbon were placed in a 5.00-L container and heated, the equilibrium concentration of CO was found to be 0.060 M. What is the equilibrium constant, Kc, for this reaction?
A) 0.010
B) 0.072
C) 0.090
D) 0.17
E) 1.2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon monoxide and chlorine combine in an equilibrium reaction to produce the highly toxic product, phosgene (COCl2) CO(g) + Cl2(g) ![Carbon monoxide and chlorine combine in an equilibrium reaction to produce the highly toxic product, phosgene (COCl<sub>2</sub>) CO(g) + Cl<sub>2</sub>(g) <sub> </sub> COCl<sub>2</sub>(g) If the equilibrium constant for this reaction is K<sub>c</sub> = 248, predict, if possible, what will happen when the reactants and product are combined with the concentrations shown. [CO] = [Cl<sub>2</sub>] = 0.010 M; [COCl<sub>2</sub>] = 0.070 M A) The reaction will proceed to the right. B) The reaction will proceed to the left. C) The reaction is at equilibrium, and no change in concentrations will occur. D) The container volume needs to be specified before a prediction can be made. E) The temperature needs to be specified before a prediction can be made.](https://d2lvgg3v3hfg70.cloudfront.net/TB5833/11ea8ef8_2b81_29d6_ab0a_897fdd8513da_TB5833_11.jpg) COCl2(g)
If the equilibrium constant for this reaction is Kc = 248, predict, if possible, what will happen when the reactants and product are combined with the concentrations shown.
[CO] = [Cl2] = 0.010 M; [COCl2] = 0.070 M
COCl2(g)
If the equilibrium constant for this reaction is Kc = 248, predict, if possible, what will happen when the reactants and product are combined with the concentrations shown.
[CO] = [Cl2] = 0.010 M; [COCl2] = 0.070 M
A) The reaction will proceed to the right.
B) The reaction will proceed to the left.
C) The reaction is at equilibrium, and no change in concentrations will occur.
D) The container volume needs to be specified before a prediction can be made.
E) The temperature needs to be specified before a prediction can be made.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 104
Related Exams