A) Higher molecular weight
B) Lower molecular weight
C) Lower wavenumbers
D) Higher wavenumbers
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the IR below and classify the compound. 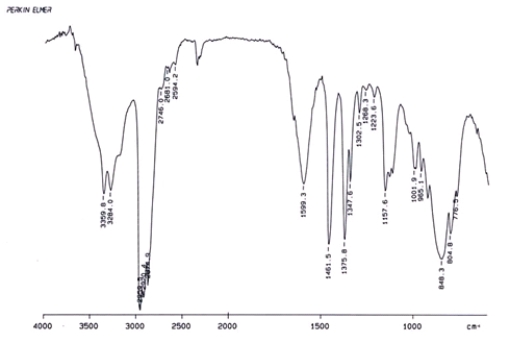
A) Alcohol
B) Aldehyde
C) Amine
D) Ketone
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An alkyne C-H bond absorbs at higher wave number than an alkene C-H bond. What does this tell you about the strength of these two bonds?
A) The alkene C-H bond is stronger.
B) The alkyne C-H bond is stronger.
C) The alkene C-C bond is stronger.
D) The alkyne C-C bond is stronger.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is (are) true about a compound that has a molecular ion peak in its mass spectrum at mass 94, and shows prominent peaks in its IR spectrum at 3600-3200 and 1600 cm-1?
A) The compound has a molecular mass of 94.
B) The compound contains a C=O group and Csp3-H hybridized bonds.
C) The compound contains an OH group and a benzene ring.
D) Both A (The compound has a molecular mass of 94) and B (The compound contains a C=O group and Csp3-H hybridized bonds) are true statements.
E) Both A (The compound has a molecular mass of 94) and C (The compound contains an OH group and a benzene ring) are true statements.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type(s) of molecular motion is (are) observed using infrared spectroscopy?
A) Stretching and bending
B) Rotation and excitation
C) Spin flipping
D) Fragmentation
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following bonds is IR inactive? 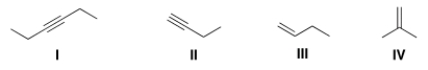
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of feature will be observed in the EI mass spectrum of chlorobenzene?
A) An absorption at 3100 cm-1
B) A signal at 113.
C) A signal at 112 and a signal at 114 in a ratio of 3:1
D) A signal at 112 and a signal at 114 in a ratio of 1:1
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a typical mass spectrum, a smaller signal is observed at a mass 1 amu higher than the molecular ion peak. Why?
A) Due to small impurities in the sample
B) Machine error
C) Because a small percentage of the compound will have a carbon that is the isotope 13C instead of 12C
D) Because of fragmentation
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An IR spectrum has the following potentially important absorptions: 3091, 3067, 2963, 2921, 2252, 1603, 1499, 1455, 1416, 1078, 1031, 941, 735, and 696 cm-1. Indicate which structure corresponds to the IR data. 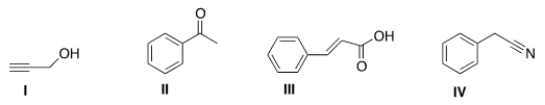
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In electron impact mass spectrometry (EIMS) , what is being detected?
A) The molecular mass of the compound
B) The molecular formula of the compound
C) The mass to charge ratio of any ionic species
D) The mass to charge ratio of any neutral species
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
13C NMR is a technique in which the total number of signals represents the number of unique carbon atoms in a molecule. Propose a structure that is consistent with the following data.
(a) The IR includes peaks at 1603 and 1495 cm-1.
(b) The 13C NMR has a total of 7 signals.
(c) The compound has one acidic proton. 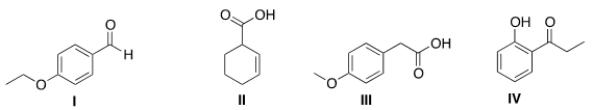
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds will have the lowest wavenumber for carbonyl absorption? 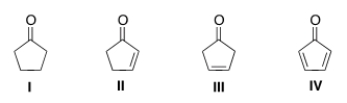
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of signal(s) would you observe in the mass and (or) infrared spectrum of the following compound? 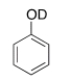
A) A signal at 95 amu
B) A signal at 94 amu
C) Two signals at 95 and 94 amu
D) A signal at 3600-3200 cm-1
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound X shows a molecular ion peak at m/z 72 in its mass spectrum, and a strong peak at ~1715 cm-1 in its IR spectrum. Which structures are possible for compound X? 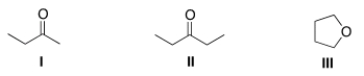
A) I
B) II
C) III
D) I and II
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the IR below and classify the compound. 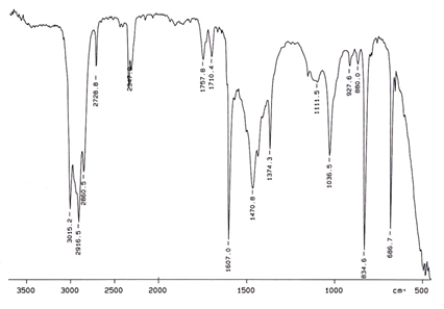
A) Alcohol
B) Arene
C) Amine
D) Ketone
E) Carbocylic acid
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about the base peak of a mass spectrum is always true?
A) The base peak corresponds to the molecular ion.
B) The base peak corresponds to the most abundant ion.
C) The base peak corresponds to the lowest m/z.
D) None of the above.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is (are) true about the IR spectrum of the compound drawn below? 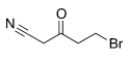
A) It shows absorptions at 3000-3150 cm-1 and 1720 cm-1.
B) It shows absorptions at 3000-2850 cm-1 and 2150 cm-1.
C) It shows absorptions at 2250 cm-1 and 1650 cm-1.
D) It shows absorptions at 2250 cm-1 and 1720 cm-1.
E) Both statements B (It shows absorptions at 3000-2850 cm-1 and 2250 cm-1) and D (It shows absorptions at 2250 cm-1 and 1720 cm-1) are true.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the indicated C-H bonds absorbs at the lowest wave number in the IR spectrum? 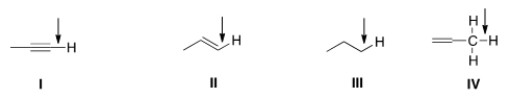
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the phenol shown below is treated with KOH, it forms a product whose IR spectrum does not show an absorption in the 3200-3600 cm-1 region. Propose a structure for the product. 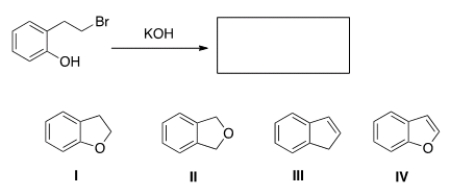
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the IR below and classify the compound. 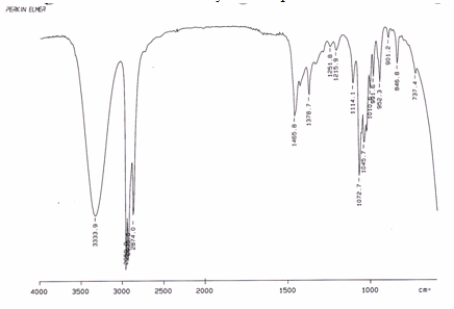
A) Alkane
B) Carboxylic acid
C) Alcohol
D) Alkene
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 42
Related Exams