A) 225 K
B) 218 K
C) 55 K
D) 273 K
E) 328 K
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify each of the following transformations as a chemical or physical change -baking a cake
A) physical
B) chemical
D) undefined
Correct Answer

verified
Correct Answer
verified
Short Answer
The simplest type of pure substance is an __________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
On a hot day, the thermometer read 95 °F. What is the temperature in degrees Celsius?
A) 77 °C
B) 113 °C
C) 35 °C
D) 63 °C
E) 178 °C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the physical state(s) corresponding to the regions on the cooling curve below. 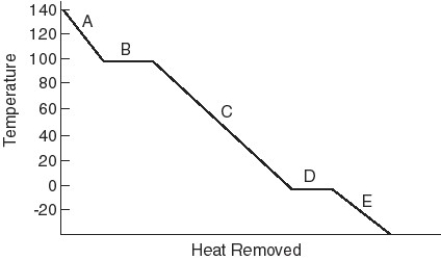 -E
A) solid and gas
B) liquid and gas
C) gas
D) liquid and solid
E) solid
F) liquid
-E
A) solid and gas
B) liquid and gas
C) gas
D) liquid and solid
E) solid
F) liquid
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify each of the following transformations as a chemical or physical change -placing photographs in a scrapbook
A) physical
B) chemical
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of calories needed to raise the temperature of 32 g of water from 12 °C to 54 °C is
A) 384 cal.
B) 1.3 cal.
C) 1300 cal.
D) 1700 cal.
E) 0.76 cal.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Bromine (Br₂) has a freezing point of -7 °C, and a boiling point of 60 °C. Indicate the state or change of state occurring at each temperature. --7 °C
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would NOT be a physical change?
A) freezing water to make ice cubes
B) tearing a piece of aluminum foil
C) boiling water for soup
D) burning gasoline in a lawnmower
E) melting gold to make jewelry
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy stored in the chemical bonds of a carbohydrate molecule is
A) specific heat.
B) kinetic energy.
C) potential energy.
D) work.
E) a calorie.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a gas, the distance between the particles is
A) very close relative to the size of the molecules.
B) close relative to the size of the molecules.
C) fixed relative to the size of the molecules.
D) small relative to the size of the molecules.
E) very large relative to the size of the molecules.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat of copper is 0.0920 cal/g °C, and the specific heat of silver is 0.0562 cal/g °C. If 100 cal of heat is added to one g of each metal at 25 °C, what is the expected result?
A) The copper will reach a higher temperature.
B) The silver will reach a higher temperature.
C) The two samples will reach the same temperature.
D) The copper will reach a temperature lower than 25 °C.
E) The silver will soften.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties describes a liquid?
A) has its own shape
B) particles are close together and move randomly
C) particles move very rapidly
D) fills the entire volume of the container
E) There is essentially no interaction between the particles.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cheeseburger from a fast food restaurant contains 19 g of fat, 20 g of carbohydrate, and 28 g of protein. How many kcal of energy does the cheeseburger contain? (The accepted caloric values for foods are 4.0 kcal/g for carbohydrate, 9 kcal/g for fat, and 4 kcal/g for protein.) Report the answer to 2 significant figures.
A) 70. kcal
B) 360 kcal
C) 17 kcal
D) 630 kcal
E) 280 kcal
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a property of a solid?
A) It takes the shape of the container.
B) It fills the volume of the container.
C) The particles move at a rapid rate.
D) The interactions between its particles are very weak.
E) The particles have fixed positions and are very close together.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Match the state of matter with each of the following descriptions of a substance. -This material has a definite volume, and a definite shape.
A) solid
B) liquid
C) solid + liquid
D) liquid + gas
E) gas
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A burn from steam at 100 °C is expected to be more severe than a burn from boiling water at 100 °C because
A) the steam is hotter than the boiling water.
B) there is more steam than water.
C) the steam will give off a large amount of heat as it condenses.
D) you are more likely to come into contact with the steam than with the boiling water.
E) All of these answers are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A kilocalorie of heat is required to raise the temperature of
A) 1 g of water from 14 °C to 15 °C.
B) 1 g of water by 10 °C.
C) 10 g of water by 10 °C.
D) 100 g of water by 10 °C.
E) 100 g of water by 100 °C.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the physical state(s) corresponding to the regions on the cooling curve below.  -B
A) solid and gas
B) liquid and gas
C) gas
D) liquid and solid
E) solid
F) liquid
-B
A) solid and gas
B) liquid and gas
C) gas
D) liquid and solid
E) solid
F) liquid
Correct Answer

verified
Correct Answer
verified
Short Answer
Bromine (Br₂) has a freezing point of -7 °C, and a boiling point of 60 °C. Indicate the state or change of state occurring at each temperature. -30 °C
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 100
Related Exams