A) x will increase and y will decrease
B) both x and y will decrease
C) x will decrease and y will increase
D) both x and y will increase
E) the changes in x and y depend on the dimension z
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system undergoes an adiabatic process in which its internal energy increases by 20 J. Which of the following statements is true?
A) 20 J of work was done on the system
B) 20 J of work was done by the system
C) the system received 20 J of energy as heat
D) the system lost 20 J of energy as heat
E) none of the above are true
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A Kelvin thermometer and a Fahrenheit thermometer both give the same reading for a certain sample. The corresponding Celsius temperature is:
A) 574 C
B) 232 C
C) 301 C
D) 614 C
E) 276 C
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gram of distilled water at 4 C:
A) will increase slightly in weight when heated to 6 C
B) will decrease slightly in weight when heated to 6 C
C) will increase slightly in volume when heated to 6 C
D) will decrease slightly in volume when heated to 6 C
E) will not change in either volume or weight
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When a certain constant volume gas thermometer is in thermal contact with water at its triple point (273.16 K) the pressure is 6.30 *104 Pa. For this thermometer a kelvin corresponds to a change in pressure of about:
A) 4.34 * 102 Pa
B) 2.31*102 Pa
C) 1.72* 103 Pa
D) 2.31 * 103 Pa
E) 1.72 * 107 Pa
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The units of thermal conductivity might be:
A) cal . cm/(s .C )
B) cal/(cm . s .C )
C) cal .s/(cm .C )
D) cm . s .C /cal
E) C /(cal . cm . s)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat capacity of object B is twice that of object A. Initially A is at 300 K and B is at 450 K. They are placed in thermal contact and the combination is isolated. The final temperature of both objects is:
A) 200 K
B) 300 K
C) 400 K
D) 450 K
E) 600 K
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Heat has the same units as:
A) temperature
B) work
C) energy/time
D) heat capacity
E) energy/volume
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy given off by 300 grams of an alloy as it cools through 50 C raises the temperature of 300 grams of water from 30 C to 40 C. The specific heat of the alloy (in cal/g .C˚) is:
A) 0.015
B) 0.10
C) 0.15
D) 0.20
E) 0.50
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The zeroth law of thermodynamics allows us to define
A) work
B) pressure
C) temperature
D) thermal equilibrium
E) internal energy
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following which might NOT vanish over one cycle of a cyclic process?
A) the change in the internal energy of the substance
B) the change in pressure of the substance
C) the work done by the substance
D) the change in the volume of the substance
E) the change in the temperature of the substance
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate of heat flow through a slab is Pcond. If the slab thickness is doubled, its cross-sectional area is halved, and the temperature difference across it is doubled, then the rate of heat flow becomes:
A) 2Pcond
B) Pcond/2
C) Pcond
D) Pcond/8
E) 8Pcond
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formation of ice from water is accompanied by:
A) absorption of energy as heat
B) temperature increase
C) decrease in volume
D) an evolution of heat
E) temperature decrease
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A constant-volume gas thermometer is used to measure the temperature of an object. When the thermometer is in contact with water at its triple point (273.16 K) the pressure in the thermometer is 8.500 * 104 Pa. When it is in contact with the object the pressure is 9.650 *104 Pa. The temperature of the object is:
A) 37.0 K
B) 241 K
C) 310 K
D) 314 K
E) 2020 K
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Possible units for the coefficient of volume expansion are:
A) mm/C
B) mm3/C
C) (C ) 3
D) 1/(C ) 3
E) 1/C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal sample of mass M requires a power input P to just remain molten. When the heater is turned off, the metal solidifies in a time T. The specific latent heat of fusion of this metal is:
A) P/MT
B) T/PM
C) PM/T
D) PMT
E) PT/M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The diagram shows four thermometers, labeled W, X, Y, and Z. The freezing and boiling points of water are indicated. Rank the thermometers according to the size of a degree on their scales, smallest to largest. 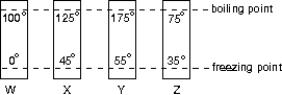
A) W, X, Y, Z
B) Z, Y, X, W
C) Z, Y, W, X
D) Z, X, W, Y
E) W, Y, Z, X
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
There is a temperature at which the reading on the Kelvin scale is numerically:
A) equal to that on the Celsius scale
B) lower than that on the Celsius scale
C) equal to that on the Fahrenheit scale
D) less than zero
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heat capacity at constant volume and the heat capacity at constant pressure have different values because:
A) heat increases the internal energy at constant volume but not at constant pressure
B) heat increases the internal energy at constant pressure but not at constant volume
C) the system does work at constant volume but not at constant pressure
D) the system does work at constant pressure but not at constant volume
E) the system does more work at constant volume than at constant pressure
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A balloon is filled with cold air and placed in a warm room. It is NOT in thermal equilibrium with the air of the room until
A) it rises to the ceiling
B) it sinks to the floor
C) it stops expanding
D) it starts to contract
E) none of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 82
Related Exams