A) sp2, 1200
B) sp, 1800
C) sp3, 109.50
D) sp3, 1200
E) sp, 1200
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements best describes the C-Cl bond in the following compound? 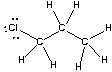
A) nonpolar; no dipole
B) polar; + at carbon and - at chlorine
C) polar; - at carbon and + at chlorine
D) ionic
E) None of these
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct Lewis structure for COCl2? 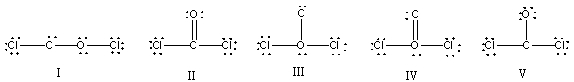
A) I
B) II
C) III
D) IV
E) V
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bonding pattern of oxygen with a formal charge of -1 could be described as:
A) One lone pair of electrons and three single bonds
B) Two lone pairs of electrons and two single bonds
C) Three lone pairs of electrons, and one single bond
D) One lone pair of electrons, one single, and one double bond
E) Zero lone pairs, and two single and one double bond
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct Lewis structure for hydrocyanic acid (HCN) including the formal charges, if any? 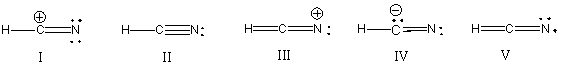
A) I
B) II
C) III
D) IV
E) V
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has two lone pairs on the central atom?
A) CO2
B) SCl2
C) NF3
D) CS2
E) SO3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound does not have a linear molecular geometry?
A) CO2
B) H2O
C) HCl
D) HCN
E) C2H2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Following is the structure for Propranolol, and antihypertensive drug. What are the hybridization state, molecular geometry and approximate bond angle at the indicated nitrogen and oxygen atom in Propranolol? 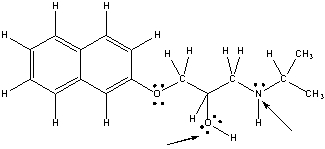
Correct Answer

verified
N: sp3, trigonal pyra...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Amino acids are building blocks of proteins. Which statement best describes the physical properties of the following amino acid? 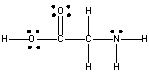
A) high melting points and low solubility in water
B) large dipole moments and no hydrogen bonding
C) high melting points and large dipole moments
D) low solubility in water and small dipole moments
E) small dipole moments and are hydrophobic
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization state and molecular geometry around sulfur atom in SOCl2?
A) sp2, tetrahedral
B) sp2, trigonal planar
C) sp3, tetrahedral
D) sp3, trigonal pyramidal
E) sp2, trigonal pyramidal
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry at the central atom in CH2Br2?
A) trigonal planar
B) trigonal pyramidal
C) sqaure planar
D) tetrahedral
E) None of these
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the formal charges on boron and oxygen in the following structure? 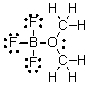
A) B = 1-, O = 1-
B) B = 1-, O = 1+
C) B = 1+, O = 1+
D) B = 1+, O = 1-
E) B = 1-, O = 0
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct Lewis structure for PH3? 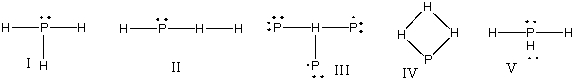
A) I
B) II
C) III
D) IV
E) V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The bond that is part of C=C in the following compound results from the overlap of which orbitals? 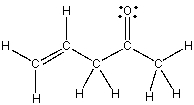
A) sp-sp2
B) sp-sp3
C) sp2-sp2
D) sp2-sp3
E) sp3-sp2
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
For the following compound identify the polar covalent bonds and indicate the direction of dipole moment using + and -. 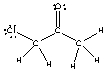
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element has the following electronic configuration? 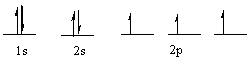
A) boron
B) carbon
C) silicon
D) nitrogen
E) fluorine
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the correct representation of partial charges at the indicated atoms? 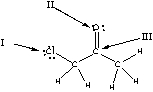
A) I = +; II = +; III = +
B) I = -; II = -; III = -
C) I = +; II = +; III = -
D) I = -; II = -; III = +
E) I = +; II = -; III = +
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Draw Lewis structure for NH2CN including formal charges, if any?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In ammonium ion, nitrogen has a valence of 4, and zero nonbonding electrons. What is the correct formal charge of nitrogen with 4 covalent bonds?
A) 2-
B) 2+
C) 1-
D) 1+
E) 0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures have 1+ formal charge on the central atom? 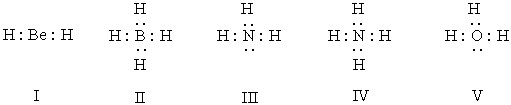
A) I
B) II
C) III
D) III & V
E) IV & V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 191
Related Exams