A) forms bonds to two metal ions.
B) has a charge of 2+ or 2-.
C) forms complex ions with a charge of 2+ or 2-.
D) has two donor atoms.
E) has medical uses.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound K2[Co(en) Cl4],the coordination number (C.N.) and oxidation number (O.N.) of cobalt are,respectively,
A) C.N.= 6; O.N.= +2.
B) C.N.= 6; O.N.= +3.
C) C.N.= 5; O.N.= +2.
D) C.N.= 5; O.N.= +4.
E) C.N.= 4; O.N.= +3.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in the complex ion [Mn(CN) 6]3-?
A) 0
B) 1
C) 2
D) 3
E) 5
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be diamagnetic?
A) Ni2+
B) Cr2+
C) Mn2+
D) Co3+
E) Ti4+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the compound K[Co(C2O4) 2(H2O) 2] (where C2O42- = oxalate) the oxidation number and coordination number of cobalt are _____ and _____,respectively.
A) -1; 4
B) -1; 6
C) +3; 4
D) +3; 6
E) +1; 6
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands has two different types of donor atoms?
A) NH3
B) H2O
C) NH4+
D) NO2-
E) ethylenediamine
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the coordination number of cobalt in the complex ion [Co(en) Cl4]-? (en = ethylenediamine)
A) 1
B) 2
C) 4
D) 6
E) 8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Why is the +2 oxidation state so common among transition elements?
Correct Answer

verified
The outermost electr...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the name of the molecules or ions that surround the metal in a complex ion?
A) Chelaters
B) Donors
C) Acceptors
D) Coordinators
E) Ligands
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these diagrams represent the crystal field splitting between d orbitals in an octahedral complex?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
Ethylenediaminetetraacetic acid (EDTA)is an effective antidote for heavy metal poisoning (e.g.,Pb2+ and Hg2+).
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following energy-level diagram could correspond to which coordination compound? 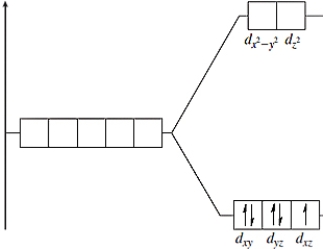
A) hexabromomanganate(II)
B) hexacyanoferrate(II)
C) hexacyanoferrate(III)
D) hexabromomanganate(IV)
E) hexachlorochromate(II)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these species is a bidentate ligand?
A) CN-
B) NH3
C) CO
D) H2NCH2CH2NH2
E) EDTA
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for pentaamminechlorocobalt(III) chloride?
A) [Co(NH3) 5Cl]Cl
B) [Co(NH3) 5Cl]Cl2
C) [Co(NH3) 5Cl]Cl3
D) [Co(NH3) 5Cl]Cl4
E) [CoCl3](NH3) 5
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which are the most common transition metals?
A) The transition metals in the 3rd period.
B) The transition metals in the 4th period.
C) The elements in Group 6B.
D) The transition metals in the 5th period.
E) The transition metals in the 6th period.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
The compound Rh(CO) (H) (PH3)2 forms cis and trans isomers.Use this information to predict the geometry of this complex isomer.
Correct Answer

verified
The complex has four ligands a...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the electron configuration of a ground-state Nb2+ ion?
A) 1s22s22p63s23p64s23d104p65s24d1
B) 1s22s22p63s23p64s23d104p64d3
C) 1s22s22p63s23p64s23d104p65s14d2
D) 1s22s22p63s23p64s23d104p64d5
E) 1s22s22p63s23p64s23d104p65s24d3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
In a reaction between a metal ion and a group of anions or a polar molecule,the anions or polar molecule acts as a _______ ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many 3d electrons does a ground-state Pd2+ ion have?
A) 8
B) 10
C) 9
D) 7
E) 6
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of structure is exhibited by most of the transition metals?
A) Close-packed
B) Tetrahedral
C) Trigonal planar
D) Trigonal pyramidal
E) None of the answers is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 128
Related Exams