A) ΔG = -RT lnK
B) ΔG = RT lnK
C) ΔGo = -RT lnK
D) ΔGo = -RT lnQ
E) ΔGo = RT lnQ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Stearic acid,nature's most common fatty acid,dimerizes when dissolved in hexane: 2C17H35COOH  (C17H35COOH) 2; ΔH°rxn = -172 kJ/mol
The equilibrium constant for this reaction at 28°C is 2.9 × 103.Estimate the equilibrium constant at 38°C.(R = 8.314 J/K• mol)
(C17H35COOH) 2; ΔH°rxn = -172 kJ/mol
The equilibrium constant for this reaction at 28°C is 2.9 × 103.Estimate the equilibrium constant at 38°C.(R = 8.314 J/K• mol)
A) 4.7 × 105
B) 2.6 × 104
C) 1.9 × 103
D) 3.2 × 102
E) 18
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider this reaction at equilibrium at a total pressure: 2H2O(g) + O2(g)  2H2O2(g)
Suppose the volume of this system is twice its initial volume and then equilibrium is reestablished.The new equilibrium total pressure will be
2H2O2(g)
Suppose the volume of this system is twice its initial volume and then equilibrium is reestablished.The new equilibrium total pressure will be
A) more than half the total initial pressure.
B) twice the total pressure.
C) less than half the total initial pressure.
D) unchanged.
E) exactly one half of the initial total pressure.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) If Q < K,then products must be converted to reactants.
B) If Q > K,then reactants must be converted to products.
C) If Q = K,then the system is at equilibrium.
D) If Q < K,then more reactants are produced.
E) None of the answers is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the free energy change,ΔG°,for the equilibrium between hydrogen iodide,hydrogen, and iodine at 453°C? (R = 8.314 J/K• mol)
2HI(g)  H2(g) + I2(g) Kc = 0.020 at T = 453°C
H2(g) + I2(g) Kc = 0.020 at T = 453°C
A) 6.4 kJ/mol
B) 8.8 kJ/mol
C) 15 kJ/mol
D) 19 kJ/mol
E) 24 kJ/mol
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the expression that relates Kc to KP?
A) Kc = KP R/T
B) KP = Kc(RT) Δn
C) KP = Kc/(RT) Δn
D) Kc KP = RT/V
E) KP = KcP/RT
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the equilibrium A2(g)  2A(g) ,if a sample of A2(g) is placed in a sealed container and allowed to come to equilibrium,which graph best represents the relationship between Kc and time as equilibrium is established?
2A(g) ,if a sample of A2(g) is placed in a sealed container and allowed to come to equilibrium,which graph best represents the relationship between Kc and time as equilibrium is established?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the endothermic reaction A2 (g)  2A(g) ,a snapshot of an equilibrium mixture of A(g) and A2 (g) at low temperature may look as follows.(Each circle represents 1.0 mol of A atoms,and the volume of the box is 1.0 L.)
2A(g) ,a snapshot of an equilibrium mixture of A(g) and A2 (g) at low temperature may look as follows.(Each circle represents 1.0 mol of A atoms,and the volume of the box is 1.0 L.) 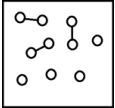 If the temperature is raised,what might the new equilibrium system look like?
If the temperature is raised,what might the new equilibrium system look like?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Solid ammonium hydrogen sulfide is introduced into a 2.00-L flask,and the flask is sealed.If this solid decomposes according to the equation NH4HS(s)  NH3(g) + H2S(g) ,KP = 0.108 at 25°C,what is the minimum mass of ammonium hydrogen sulfide that must be present in the flask initially if equilibrium is to be established at 25°C? (R = 0.08206 L • atm/K • mol)
NH3(g) + H2S(g) ,KP = 0.108 at 25°C,what is the minimum mass of ammonium hydrogen sulfide that must be present in the flask initially if equilibrium is to be established at 25°C? (R = 0.08206 L • atm/K • mol)
A) 0.917 g
B) 1.37 g
C) 2.74 g
D) 0.581 g
E) 0.452 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction at equilibrium in a reaction vessel,which one of these changes would cause the I2 concentration to increase? 2NOI(g)  2NO(g) + I2(g) ,ΔHºrxn = 45.3 kJ/mol
2NO(g) + I2(g) ,ΔHºrxn = 45.3 kJ/mol
A) Add some NO
B) Expand the gas mixture into a larger volume
C) Decrease the temperature
D) Increase the system pressure
E) Remove some NOI
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A change in a concentration will not change the position of equilibrium.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Increasing the temperature of an exothermic reaction causes the equilibrium constant to increase and shifts the equilibrium toward products.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
For any reaction,if ΔG° > 0,then K < 1.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 1500°C the equilibrium constant for the reaction CO(g) + 2H2(g)  CH3OH(g) has the value KP = 1.4 × 10-7.What is ΔG° for this reaction at 1500°C? (R = 8.314 J/K • mol)
CH3OH(g) has the value KP = 1.4 × 10-7.What is ΔG° for this reaction at 1500°C? (R = 8.314 J/K • mol)
A) 105 kJ/mol
B) 1.07 kJ/mol
C) -233 kJ/mol
D) -105 kJ/mol
E) 233 kJ/mol
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction 2X(g) + Y(g)  2Z(g) ,Kc = 1.00 ×103 at 500 K.If at equilibrium the concentration of X is 0.20 M and the concentration of Y is 0.50 M,what is the equilibrium concentration of Z?
2Z(g) ,Kc = 1.00 ×103 at 500 K.If at equilibrium the concentration of X is 0.20 M and the concentration of Y is 0.50 M,what is the equilibrium concentration of Z?
A) 2.2 M
B) 3.2 M
C) 3.5 M
D) 4.5 M
E) 7.1 M
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The equilibrium constant expression for the reaction
CuO(s)+ H2(g) ![The equilibrium constant expression for the reaction CuO(s)+ H<sub>2</sub>(g) Cu(s)+ H<sub>2</sub>O(g)is K<sub>c</sub> = [H<sub>2</sub>]/[H<sub>2</sub>O].](https://d2lvgg3v3hfg70.cloudfront.net/TB6485/11eab46b_9cb6_8903_a889_7d62405573cc_TB6485_11.jpg) Cu(s)+ H2O(g)is Kc = [H2]/[H2O].
Cu(s)+ H2O(g)is Kc = [H2]/[H2O].
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The observation that at equilibrium,the reaction quotient equals the equilibrium constant,is representative of which law?
A) Law of equal states
B) Reversibility law
C) Law of equivalence
D) Law of reactant-product equivalence
E) Law of mass action
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At a certain temperature the reaction CO2(g) + H2(g)  CO(g) + H2O(g)
Has Kc = 2.50.If 2.00 mol of carbon dioxide and 1.50 mol of hydrogen are placed in a 5.00-L vessel and equilibrium is established,what is the equilibrium concentration of carbon monoxide?
CO(g) + H2O(g)
Has Kc = 2.50.If 2.00 mol of carbon dioxide and 1.50 mol of hydrogen are placed in a 5.00-L vessel and equilibrium is established,what is the equilibrium concentration of carbon monoxide?
A) 0.209 M
B) 1.33 M
C) 0.267 M
D) 0.667 M
E) 0.600 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At 850°C,the equilibrium constant,KP,for the reaction C(s) + CO2(g)  2CO(g) has a value of 10.7
If the total pressure in the system at equilibrium is 1.000 atm,what is the partial pressure of carbon monoxide at equilibrium?
2CO(g) has a value of 10.7
If the total pressure in the system at equilibrium is 1.000 atm,what is the partial pressure of carbon monoxide at equilibrium?
A) 0.362 atm
B) 0.489 atm
C) 0.667 atm
D) 0.915 atm
E) 0.921 atm
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium carbonate,Na2CO3(s) ,may be prepared by heating sodium bicarbonate,NaHCO3(s) . 2NaHCO3(s)  Na2CO3(s) + CO2(g) + H2O(g) KP = 0.23 at 100ºC
If a sample of NaHCO3 is placed in an evacuated flask and allowed to achieve equilibrium at 100ºC,what will the total gas pressure be?
Na2CO3(s) + CO2(g) + H2O(g) KP = 0.23 at 100ºC
If a sample of NaHCO3 is placed in an evacuated flask and allowed to achieve equilibrium at 100ºC,what will the total gas pressure be?
A) 0.46 atm
B) 0.96 atm
C) 0.23 atm
D) 0.48 atm
E) 0.11 atm
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 135
Related Exams