A) N
B) S
C) He
D) Cl
E) Fe
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The elements in Group 8A are called the ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which pair of elements would be most likely to form an ionic compound?
A) P and Br
B) Zn and K
C) F and Al
D) C and S
E) Al and Rb
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the empirical formula for hexane,C6H14?
A) C12H28
B) C6H14
C) C3H7
D) CH2.3
E) C0.43H
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an ionic compound?
A) H2S
B) NH3
C) I2
D) KI
E) CCl4
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The alkali metal elements are found in _______ of the periodic table.
A) Group 1A
B) Group 2A
C) Group 3A
D) Period 7
E) Period 1
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of protons (p) ,electrons (e) ,and neutrons (n) in one atom of chlorine- 37.
A) 37 p,37 e,17 n
B) 17 p,17 e,37 n
C) 17 p,17 e,20 n
D) 37 p,17 e,20 n
E) 17 p,37 e,17 n
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the number of electrons and identify the correct symbol for an atom with 17 protons and 18 neutrons.
A) 17 electrons, ![]()
B) 18 electrons, ![]()
C) 17 electrons, ![]()
D) 17 electrons, ![]()
E) 18 electrons, ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What terms defines a mass which is exactly equal to 1/12 the mass of one carbon-12 atom?
A) Isotope number
B) Mass number
C) Mass-to-charge ratio
D) Atomic number
E) Atomic mass unit
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metal?
A) Nitrogen,N,Z = 7
B) Phosphorus,P,Z = 15
C) Arsenic,As,Z = 33
D) Thallium,Tl,Z = 81
E) Silicon,Si,Z = 14
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The mass of a neutron is equal to the mass of a proton plus the mass of an electron.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lithium forms compounds which are used in dry cells,storage batteries,and in high-temperature lubricants.It has two naturally occurring isotopes,6Li (isotopic mass = 6.015123 amu) and 7Li (isotopic mass = 7.016005 amu) .Lithium has an atomic mass of 6.9412 amu.What is the percent abundance of lithium-6?
A) 92.53%
B) 86.65%
C) 49.47%
D) 7.47%
E) 6.015%
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The chemical formula for iron(II) nitrate is
A) Fe2(NO3) 3.
B) Ir(NO2) 2.
C) Fe2N3.
D) Fe(NO3) 2.
E) Fe(NO2) 2.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When J.J.Thomson discovered the electron,what physical property of the electron did he measure?
A) its charge,e
B) its charge-to-mass ratio,e/m
C) its temperature,T
D) its mass,m
E) its atomic number,Z
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the number of protons,electrons,and neutrons for the isotope gold-118.The symbol for gold is Au.
A) 118 protons,118 electrons,79 neutrons
B) 79 protons,79 electrons,118 neutrons
C) 79 protons,79 electrons,39 neutrons
D) 118 protons,118 electrons,39 neutrons
E) 79 protons,39 electrons,118 neutrons
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is represented by X in the atomic symbol notation 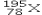 ?
?
A) Iridium
B) Platinum
C) Palladium
D) Selenium
E) Magnesium
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for lead(II) oxide?
A) PbO
B) PbO2
C) Pb2O
D) PbO4
E) Pb2O3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of Ba(NO2) 2·3H2O?
A) barium nitrite
B) trihydrobarium(II) nitrite
C) barium nitrite trihydrate
D) barium(II) nitrite trihydrate
E) barium nitrate trihydrate
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The elements in a column of the periodic table are known as
A) metalloids.
B) a period.
C) noble gases.
D) a group.
E) nonmetals.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the number of protons and identify the correct symbol for an atom with 20 neutrons and 20 electrons.
A) 20 protons, ![]()
B) 20 protons, ![]()
C) 20 protons, ![]()
D) 40 protons, ![]()
E) 40 protons, ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 120
Related Exams